10. heterocicli
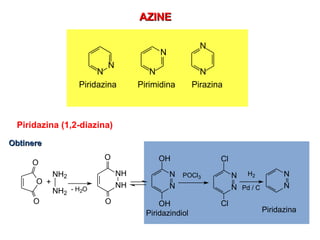
- 1. AAZZIINNEE N N N N N N Piridazina Pirimidina Pirazina Piridazina (1,2-diazina) OObbttiinneerree POCl3 N N Cl Cl N H2 Pd / C N Piridazina O O O NH O NH2 NH2 - H2O NH O OH N N OH + Piridazindiol
- 2. O O O NH2 NH NH2 - H2O NH O O OH N N OH + NH NH O O OH N N OH N N O O N N O O + Sint.Dien [O] 1,4-Ftalazindiona ftalhidrazida [O] = hipoclorit de tert-butil, tetraacetat de Pb
- 3. Pirimidina (1,3-diazina) O C NH2 NH2 ROOC CH2 ROOC EtO - 2 ROH O HN O NH O N OH HO N OH + Uree Ester malonic Acid barbituric taut. OObbttiinneerree N OH POCl3 N HO N OH Cl Cl N Cl Zn, H2O N N Pirimidina
- 4. Derivaţi ai pirimidinei Uracil (2,4-dihidroxi-pirimidina) ROOC O C EtO CH2 OHC NH2 NH2 N OH HO N + ROH + H2O Uracil + Uree Ester formilacetic Citosina (2-hidroxi-4-amino-pirimidina) POCl3 N Cl C N 2H5S NH3 N NH2 C N 2H5S H2O HCl NH2 N N HO ROOC CH2 OHC EtO N N C2H5S OH -ROH + Etil-tiouree Ester formilacetic C NH NH2 C2H5S -H2O Citosina
- 5. 2-Amino-pirimidina ROOC CH2 OHC EtO N OH H2N N -ROH - H2O C NH NH2 H2N + POCl3 N Cl H2N N N red. H2N N 2-Amino-pirimidina N N N N N N N N N N Reactivitate Caracter aromatic – derivaţii substituiţi la nucleu cu grupe aril, halogen, alchil şi nitro - scade pe măsură ce se introduc la nucleu grupe –OH şi -NH2 - substituţia electrofilă decurge greu
- 6. ROOC + + 2 ROH C ROOC Et Et EtO C NH2 O NH2 NH NH O O Et Et O Veronal
- 7. Pirazina (1,4- diazina) N N N .. .. N N N N N N .. .. .. .. .. N .. .. N N .. R C=O R CH NH2 H2N CH R O=C N R R R N R N R R R N R R + 2,3,5,6-tetra-alchilpirazine OObbttiinneerree
- 8. N N N N N N N N 2,5-dimetilpirazina 2,3,5,6-tetrametilpirazina 2-metil-3-etilpirazina 2-acetilpirazina nuci prăjite N N nuci prăjite cafea prăjită NH COCH3 COCH3 popcorn COCH3 N COCH3 2-acetil-3-etilpirazina 2-acetil-1,4,5,6-tetrahidropiridina 2-acetilpirol cartofi copti
- 9. PPUU RRIINNEE 6 7 N 5 N N N H 1 2 3 4 8 9
- 10. Obţinere Sinteza Baeyer-Fischer HN O O NH O HNO2 HN O O O NH NO 4[H] HN O O O NH NH2 HNCO HN O O NH O NH CO NH2 Acid barbituric Uramil -H2O N N N N OH H HO OH Acid uric POCl3 N N N N Cl H Cl Cl red. N N N N Purina H
- 11. Sinteza Traube ROOC C NC H H O EtO H - ROH H C NH2 O NH2 N OH NH NH2 HO N NH O NH 6-Aminouracil + Uree Ester cianacetic HNO2 N OH HO N NO NH2 red. N OH HO N NH2 NH2 5,6-Diaminouracil OH NH N - ROH HO N COOR NH2 Cl-COOR N N Acid uric N N OH H HO OH ROOC C NC H H EtO C NH2 S NH2 N OH HS N NH2 NH -ROH H NH S O H NH 6-Aminotiouracil + Tiouree HNO2 N OH HS N NO NH2 OH red. N HS N NH2 NH2 OH N N N NH Hipoxantina HCOOH N HNO3 N N NH HS OH
- 12. ROOC C EtO C NH2 NC H H HN O H H NH2 N OH H2N N NH2 NH NH NH HN -ROH N N N N H NH2 HCOOH H2N OH 1. HNO2 2. red. N OH H2N N NH2 Guanina Guanidina NC C NC HH C EtO NH2 S NH H H NH2 N NH2 NH HS N NH2 NH S NH 1. HNO2 2. red. N NH2 HS N NH2 HCOOH N NH2 N N N H HS NH2 HNO3 N N N N H NH2 Tiouree Dinitrilmalonic Adenina
- 13. N N N N H HO OH OH POCl3 3HI N N N N N H Cl Cl Cl N N N H EtO-Na+ KOH NH3 N N N N H OEt EtO Cl N N N N H OH HO HI N N N N H OH Cl Cl HI N N N N H OH 1. NH3 2. HI N N N N H OH H2N N N N N H Cl NH2 Cl HI N N N N H NH2 Adenina Guanina Hipoxantina Xantina
- 14. NH2 N N N N HO O CH2 OH OH O P ATP O O O P P OH O OH O OH HO OH N O CONH2 O O CH2 O OH OH OH CH2 O P O O P O Codehidraza I (DNP) N N NH2 N N