Platelet Disorders: Causes and Diagnosis
- 1. Overview of Disorders of Platelets Vidur Singh
- 2. Haemostasis • Hemostasis is a dynamic process in which the platelet and the blood vessel wall play key roles. • Platelets become activated upon adhesion to von Willebrand factor (vWF) and collagen in the exposed subendothelium after injury. • Platelet activation is also mediated through shear forces imposed by blood flow itself, particularly in areas where the vessel wall is diseased, and is also affected by the inflammatory state of the endothelium. • The activated platelet surface provides the major physiologic site for coagulation factor activation, which results in further platelet activation and fibrin formation. Genetic and acquired influences on the platelet and vessel wall, as well as on the coagulation and fibrinolytic systems, determine whether normal hemostasis, or bleeding or clotting symptoms, will result.
- 6. Prothrombotic effects of endothelial cells Antithrombotic effects of endothelial cells
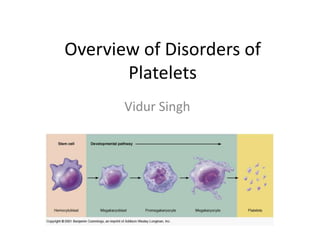
- 8. Platelets (*) • (*) are released from the megakaryocytes, likely under the influence of flow in the capillary sinuses. • Main regulator of (*)production is the hormone thrombopoietin (TPO), which is synthesized in the liver. • Normal BLOOD platelet count = 150,000–450,000/L. • (*) synthesis increases with inflammation and specifically by interleukin 6.
- 9. Platelets • Are very active, aneucleate and they have limited capacity to synthesize new proteins • Circulate with an average life span of 7–10 days. • Approximately 1/3 of the platelets reside in the spleen, and this number increases in proportion to splenic size, although the platelet count rarely decreases to <40,000/L as the spleen enlarges.
- 10. THROMBOPOIETIN (TPO) AND PLATELET PRODUCTION INTERACTION • TPO binds to receptor on • HENCE, a reduction in platelets and platelet and megakaryocytes • removed from the megakaryocyte mass circulation. • increases the level of TPO, which then stimulates platelet production.
- 11. Thrombopoietin
- 13. Functional Disorders (Qualitative) Disorders of platelets Disorders in the number of platelets
- 14. Diagnostic Tools • Peripheral blood smears • Platelet-associated immunoglobulin G • In vitro platelet function analyzer 100 • Platelet aggregation • Imaging studies • Bone marrow examination
- 15. Disorders that cause Thrombocytopenia (lower number of platelets, than the normal range of platelet count, in the blood)
- 16. Thrombocytopenias • Thrombocytopenia a platelet count of less than 150,000/mm3 (<150,000/mm3) • N.B. With normal platelet function, thrombocytopenia is rarely the cause of bleeding unless the count is less than 50,000/mm3. • Thrombocytopenia should always be confirmed by examination of a peripheral smear. • It can be caused by: – decreased platelet production – increased destruction – Sequestration (DISORDERS OF DISTRIBUTION of platelets) – or a combination of these causes.
- 17. Thromboyctopenia
- 18. vitamin deficiency (e.g., folate, vitamin B12) Marrow infiltration by Storage diseases tumor Infections Drugs (frequently (including HIV) chemotherapeutic agents or alcohol, but Causes of other medications in platelet rare cases) underproduction Marrow failure radiotherapy syndromes (e.g. aplastic anemia)
- 19. Types of thrombocytopenias Infection-Induced Thrombocytopenia Drug-Induced Thrombocytopenia Heparin-Induced Thrombocytopenia Immune Thrombocytopenic Purpura (ITP) Inherited Thrombocytopenia Thrombotic Thrombocytopenic Purpura Hemolytic Uremic Syndrome
- 20. Differential diagnosis of thrombocytopenia. Evaluation of thrombocytopenia is best organized according to the apparent defect in platelet production, distribution, or destruction.
- 22. Platelet Underproduction • The hallmark of platelet underproduction is decreased marrow megakaryocytes or, when available, a decreased peripheral blood reticulated platelet count
- 23. Platelet Sequestration • Hypersplenism from a variety of causes, including liver disease or malignancy, may result in platelet sequestration. • Mild to moderate thrombocytopenia is caused by platelet sequestration when there is an associated mild reduction in neutrophil count and hemoglobin and with minimal impairment of hematopoiesis on bone marrow examination. • If physical examination fails to detect splenomegaly, evaluation with ultrasonography or radionuclide imaging is recommended to document splenomegaly.
- 24. Increased Platelet Destruction • The hallmark of increased platelet destruction is increased marrow megakaryocytes or, when available, high reticulated platelet count. Platelet destruction results from various immune conditions, including the following: • Immune thrombocytopenic purpura (ITP) • Thrombotic microangiopathies • Post-transfusion purpura (PTP) • Heparin-induced thrombocytopenia (HIT) • Disseminated intravascular coagulation (DIC)
- 25. Hemolytic Uremic Syndrome • HUS is a syndrome characterized by acute renal failure, microangiopathic hemolytic anemia, and thrombocytopenia. • It is seen predominantly in children and in most cases is preceded by an episode of diarrhea, often hemorrhagic in nature. Escherichia coli O157:H7 is the most frequent, although not only, etiologic serotype. HUS not associated with diarrhea (termed DHUS) is more heterogeneous in presentation and course. • Some children who develop DHUS have been found to have mutations in genes encoding Factor H, a soluble complement regulator, and membrane cofactor protein that is mainly expressed in the kidney.
- 26. Treatment of hemolytic uremic syndrome: Treatment • Treatment of HUS is primarily supportive. In D+HUS, many (~40%) children require at least some period of support with dialysis; however, the overall mortality is <5%. In D–HUS the mortality is higher, approximately 26%. • Plasma infusion or plasma exchange has not been shown to alter the overall course. ADAMTS13 levels are generally reported to be normal in HUS, although occasionally they have been reported to be decreased. • As ADAMTS13 assays improve, they may help in defining a subset that better fits a TTP diagnosis and may respond to plasma exchange.
- 27. Pathogenesis of thrombotic thrombocytopenic purpura (TTP)
- 28. Increased Platelet Destruction • The hallmark of increased platelet destruction is increased marrow megakaryocytes or, when available, high reticulated platelet count. Platelet destruction results from various Thrombocytosis immune conditions, including the following: • Immune thrombocytopenic purpura (ITP) • Thrombotic microangiopathies • Post-transfusion purpura (PTP) • Heparin-induced thrombocytopenia (HIT) • Disseminated intravascular coagulation (DIC)
- 29. Thrombocytosis • Thrombocytosis is almost always due to: Iron deficiency; Inflammation, cancer, or infection (reactive thrombocytosis); or An underlying myeloproliferative process [essential thrombocythemia or polycythemia vera or, rarely, the 5q- myelodysplastic process. Medications that can cause reactive thrombocytosis include: – Epinephrine (Adrenalin Chloride, EpiPen) – Tretinoin – Vincristine • Patients presenting with an elevated platelet count should be evaluated for underlying inflammation or malignancy, and iron deficiency should be ruled out. • Thrombocytosis in response to acute or chronic inflammation has not been associated with an increased thrombotic risk. In fact, patients with markedly elevated platelet counts (>1.5 million), usually seen in the setting of a myeloproliferative disorder, have an increased risk of bleeding. This appears to be due, at least in part, to acquired von Willebrand disease (vWD) due to platelet-vWF adhesion and removal.
- 30. Blood smear of thrmobocytosis
- 31. Essential thrombocythemia • Essential thrombocythaemia (ET) is a condition affecting the cells in the bone marrow that make platelets [A myeloproliferative disorder], whereby there is overproduction of platelets, leading to an increased propensity to thrombosis formation and hence, blockage of blood vessels. • It is a rare condition that can affect people at any age, including children, but it's most common in people over 50. • Some people have a change (mutation) in a gene, called the JAK2 gene, that may have caused their condition. This gene produces an important protein involved in blood production. The fault in the gene occurs during the person's lifetime. It isn't inherited and can't be passed onto children. Finding this faulty gene can help to make the diagnosis.
- 32. Thrombopoietin
- 33. Treatment • Essential Thrombocytosis – Low-dose aspirin -- may treat headache and burning pain in the skin. – Hydroxyurea (Droxia, Hydrea) or anagrelide (Agrylin) -- reduces number of blood cells. – Aminocaproic acid -- reduces bleeding. This treatment may be used before surgery to prevent bleeding as well.
- 34. Signs and Symptoms of essential thrombocytosis – Heart attack or stoke – Headache – Burning or throbbing pain, redness, and swelling of the hands and feet – Bruising – Gastrointestinal bleeding or blood in the urine
- 35. Increased Platelet Destruction • The hallmark of increased platelet destruction is increased marrow megakaryocytes or, when available, high reticulated platelet count. Platelet destruction results from various Disorders of Platelet immune conditions, including the following: Function • Immune thrombocytopenic purpura (ITP) • Thrombotic microangiopathies • Post-transfusion purpura (PTP) • Heparin-induced thrombocytopenia (HIT) • Disseminated intravascular coagulation (DIC)
- 36. Congenital disorders of platelet function Von Willebrand Disease Glanzmann’s Thrombasthenia Bernard-soulier Syndrome
- 37. Gp IIb-IIIa complex on platelet surface
- 38. Gp Ib receptor on platelets
- 39. N.B. WITH BERNARD- SOULIER; the abnormal RISTO is not corrected by the addition of vWF Aggregometer studies in patients with congenital disorders of platelet function. Upper panels: The normal pattern of aggregation to ADP, epinephrine, collagen, arachidonic acid, and ristocetin. von Willebrand disease can be identified because of an abnormal ristocetin aggregation pattern. This condition is distinctly different from patterns seen with storage pool disease, aspirin inhibition, and Glanzmann thrombasthenia.
- 40. Acquired Abnormalities of Platelet Function MYELOPROLIFERATIVE DISEASE DYSPROTEINEMIA CARDIOPULMONARY BYPASS UREMIA LIVER DISEASE DRUG INHIBITION
- 41. MYELOPROLIFERATIVE DISEASE • Patients with myeloproliferative disorders (ie, polycythemia vera, myeloid metaplasia, idiopathic myelofibrosis, essential thrombocythemia, and chronic myelogenous leukemia) frequently exhibit abnormal platelet function. • Some of these patients have very high platelet counts and demonstrate either abnormal bleeding, or a tendency for arterial or venous thrombosis, or even both. Although the height of the platelet count alone does not correlate with the bleeding or thrombotic tendency, thrombocytosis in excess of 1 million/µL is considered to be a risk factor. • At the same time, increased platelet turnover, as measured by the reticulated platelet count, may correlate with thrombotic risk. In patients with polycythemia vera, expansion of the total blood volume and an increase in blood viscosity may also contribute to the thrombotic risk. • Other laboratory findings can be quite variable. The BT may be prolonged, but is a poor predictor of abnormal bleeding. Perhaps the most consistent laboratory abnormalities in bleeding patients are defects in epinephrine-induced aggregation and dense and alpha granule function. Bleeding caused by an acquired form of vWD also may be observed in these disorders, secondary to a loss of higher- molecular-weight vWF multimers.
- 42. CLINICAL FEATURES • There are no specific symptoms or signs that indicate a platelet functional defect. • Inherited defects are rare and are generally characterized by a relatively mild bleeding tendency. von Willebrand disease (vWD) is an exception to this rule, since certain vWD subtypes are associated with severe and even fatal bleeding. • Patients with platelet functional defects generally present with: Easy bruisability Mucocutaneous bleeding of a purpuric nature Bleeding from the gastrointestinal and genitourinary tracts (often with severe menorrhagia) rather than the petechial bleeding that characterizes thrombocytopenia. It is not unusual for the bleeding tendency to escape detection until aggravated by another abnormality. For example, the defect may first be suspected because of excessive bleeding following minor surgery or a dental extraction or unusual mucocutaneous bleeding following the administration of anticoagulants or a platelet inhibitor such as aspirin. Therefore, a history of unusual bleeding, a family history suggestive of a congenital abnormality, and the clinical picture can provide important clues.
- 43. CLINICAL FEATURES Bleeding gums Easy Bruising
- 44. Laboratory Studies 1. BLEEDING TIME AND TESTS FOR FACTOR DEFICIENCIES • The bleeding time (BT) has traditionally been used as a screening test for the presence of a platelet functional defect. If performed carefully in a well-standardized manner, the BT correlates with both platelet number and function. • With platelet counts greater than 100,000/µL, the BT should be less than 8 minutes. As the count falls below this level, the BT lengthens, reaching times of 20 to 25 minutes when the count falls to 10,000/µL. • Patients with functional defects, such as severe vWD, can show BTs in excess of 20 minutes with a normal platelet count. Patients with an acquired functional defect secondary to aspirin therapy or uremia show more modest prolongations of the BT (8 – 20 minutes).
- 45. Laboratory Studies 2. COMPLETE BLOOD COUNT AND BLOOD FILM • A complete blood count (CBC) with examination of the blood film can also be helpful. • The CBC can provide evidence of hematopoietic disease, especially a myeloproliferative disorder where high numbers of circulating platelets are associated with abnormal function. • Platelet morphology can help in diagnosing disorders such as Bernard-Soulier syndrome and α- granule deficiency (gray platelet syndrome).
- 46. Laboratory Studies 3. MEASUREMENTS OF PLATELET ACTIVATION/AGGREGATION • Direct measurements of platelet activation/aggregation are possible using an aggregometer or flow cytometer. • The aggregometer provides a graphic display of the wave of platelet aggregation seen in response to agonists such as ADP, epinephrine, or collagen, and the agglutination response to ristocetin. Specific functional defects respond differently to these agonists. • For example, patients with vWD specifically show decreased or absent agglutination with ristocetin [ristocetin-induced platelet agglutination assay (RIPA)], whereas other disorders such as storage pool disease demonstrate poor responses to ADP, epinephrine, and collagen.
- 47. ASSAYS FOR VON WILLEBRAND FACTOR • Full evaluation of the patient with vWD requires an array of tests, including assays for factor VIII activity, vWF antigen, vWF activity, and vWF multimer pattern by agarose gel electrophoresis. • Together with the patient's bleeding history, family history, and BT, these assays will diagnose and classify vWD into one of several clinically important subtypes.
- 48. TREATMENT for abnormalities of platelet function • Abnormalities in platelet function are often first appreciated as a complication of an acute illness or surgery and multiple aggravating factors may play a role in determining the severity of the bleeding tendency. • An accurate diagnosis is not usually easily made • Hence, treatment should address as many potential contributing factors as possible. • This list includes discontinuing drugs that inhibit platelet function, empirically replacing vWF or treating with DDAVP (Desmopressin) and, according to the severity of the patient's bleeding, transfusing normal platelets. • Although this approach lacks precision, it is effective. Both acquired and congenital disorders of platelet function can be acutely reversed in order to control severe clinical bleeding.
- 49. Treatment cont’d • Long-term management of a platelet functional defect should be based on an accurate diagnosis. Patients with congenital defects should be counseled to avoid drugs that can aggravate the functional abnormality and cause bleeding. • Obviously, aspirin and nonsteroidal analgesics are prime offenders. vWD and thrombasthenia patients demonstrate significant prolongations of the bleeding time with aspirin ingestion and are at greater risk for clinical bleeding. • These patients should also be educated regarding the nature of their abnormality and should carry identification or wear a medical alert bracelet. This information can be invaluable as a guide to appropriate transfusion therapy in an emergency situation.
- 50. Treatment Cont’d • As a general principle, the nature of the functional abnormality will guide the choice of therapy. • For example, the vWD patient who lacks normal amounts of vWF will respond to agents that increase plasma vWF levels. In this situation, the platelets will function normally once the vWF abnormality is corrected. In contrast, patients with congenital defects of platelet receptor expression, granule content, or platelet metabolism will require platelet transfusion. • As for the acquired abnormalities of platelet function, the best approach to therapy lies somewhere in between. There is clinical evidence that patients with acquired defects secondary to drug ingestion, uremia, and liver disease will respond to DDAVP, vWF replacement, or both. These data suggest that an increase in plasma vWF levels may partially compensate for a platelet-based defect.
- 51. Bibliography • http://emedicine.medscape.com/article/201722- overview#aw2aab6b9 • http://home.kku.ac.th/acamed/kanchana/bsi.htm • Kumar, V., Abbas, A. K., Fausto, N., & Mitchell, R. N. (2007). Robbin's Basic Pathology 8th Edition. Philadelphia, PA: Saunders Elsevier. • al, F. e. (2008). Harrison's Principles of Internal Medicine, 17th Edition. USA: The McGraw-Hill Companies, Inc.
Notas do Editor
- http://img.medscape.com/article/706/262/706262-fig1.jpg
- http://img.medscape.com/article/706/262/706262-fig1.jpg
- Read more: http://www.umm.edu/altmed/articles/myeloproliferative-disorders-000114.htm#ixzz2A7A2KIOv
