Covid 19 diagnosis and treatment protocol
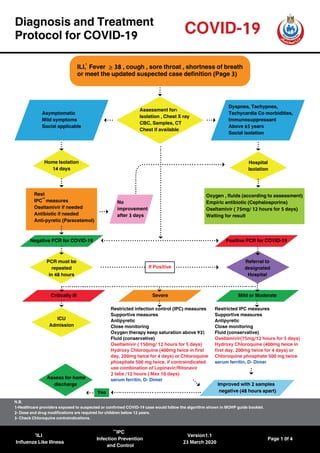
- 1. Critically ill Mild or Moderate PCR must be repeated in 48 hours Referral to designated Hospital If Positive Diagnosis and Treatment Protocol for COVID-19 COVID-19 ≥ILI, Fever 38 , cough , sore throat , shortness of breath or meet the updated suspected case definition (Page 3) Assessment for: Isolation , Chest X ray CBC, Samples, CT Chest if available Home Isolation 14 days Hospital Isolation Rest IPC measures Oseltamivir if needed Antibiotic if needed Anti-pyretic (Paracetamol) Restricted IPC measures Supportive measures Antipyretic Close monitoring Fluid (conservative) Oseltamivir(75mg/12 hours for 5 days) Hydroxy Chloroquine (400mg twice in first day, 200mg twice for 4 days) or Chloroquine phosphate 500 mg twice serum ferritin, D- Dimer Restricted infection control (IPC) measures Supportive measures Antipyretic Close monitoring Oxygen therapy keep saturation above 93% Fluid (conservative) Oseltamivir ( 150mg/ 12 hours for 5 days) Hydroxy Chloroquine (400mg twice in first day, 200mg twice for 4 days) or Chloroquine phosphate 500 mg twice, if contraindicated use combination of Lopinavir/Ritonavir 2 tabs /12 hours ( Max 10 days) serum ferritin, D- Dimer Oxygen , fluids (according to assessment) Empiric antibiotic (Cephalosporins) Oseltamivir ( 75mg/ 12 hours for 5 days) Waiting for result Dyspnea, Tachypnea, Tachycardia Co morbidities, Immunosuppressant Above 65 years Social isolation No improvement after 3 days Negative PCR for COVID-19 Positive PCR for COVID-19 Severe Asymptomatic Mild symptoms Social applicable Yes Improved with 2 samples negative (48 hours apart) ICU Admission Assess for home discharge ILI Influenza Like Illness N.B. 1-Healthcare providers exposed to suspected or confirmed COVID-19 case would follow the algorithm shown in MOHP guide booklet. 2- Dose and drug modifications are required for children below 12 years. 3- Check Chloroquine contraindications. IPC Infection Prevention and Control Version1.1 23 March 2020 Page 1 0f 4
- 2. ICU Management Protocol for COVID-19 Failed or not applicable Medications: Restrict IV fluids Antibiotics according to the guidelines Oseltamivir (150mg Twice daily) Hydroxy Chloroquine (or Chloroquine phosphate) Azithromycin (500mg daily ) Hydrocortisone (50 mg every 6 hours if hypotensive patient ) Consider Theraputic anticoagulants if D-Dimer > 2 Tocilizumab 4-8 mg/Kg maximum 400 mg once only /Or if not available methylprednisolone 2 mg/kg/24 hours for 5 days if HScore >169 Laboratory investigations: 1-PCR swab for COVID 19 every 72 hours. 2- CBC with differential daily. 3- AST, creatinine, D-dimer, Ferritin, Triglycerides, Fibrinogen, ESR, CRP daily. 4-Cultures on admission and whenever needed. Imaging: 1-Daily chest X-Ray. 2-Echocardiography with starting hemodynamic instability or inappropriate tachycardia. 3- Abdominal ultrasound if needed. Invasive Mechanical ventilation Protocol: Sedation (Midazolam or Propofol or Dexmedetomidine). Muscle paralysis (Atracurium). Consider Prone Positioning. Volume cycled control mode (VCV) start FiO2: 1 Tidal volume 4-8 ml/kg PBW. PEEP ≥ 10 cm H2 O. Respiratory rate < 30 BPM. Target Plateau Pressure < 30 cm H2 O.Switch to pressure controlled ventilation (PCV) with PIP of 30 if plateau pressure is high on VCV. Target Driving Pressure < 15 cm H2 O). Invasive Mechanical Ventilation (IMV)if: PaO2 <60mmHg or O2 saturation < 90% despite oxygen or NIV Progressive hypercapnia Respiratory acidosis (PH <7.3) Progressive or refractory Septic shock Non Invasive Ventilation trial (NIV) Conscious patient with minimal secretions Shall be short (30min). If Sp02 < 93% or hypercapnia ≥ 40 cmH2 0, PH <7.3 shift to IMV Improving Yes No Yes Improved with 2 samples negative (48 hours apart) Deteriorating or severe myocarditis Not deteriorating Keep IMV Consider ECMO Assess for weaning Weaning criteria PaO2 ≥ 60 mmHg on FiO2 < 40–50 % and PEEP ≤ 5 – 8 cm H2 O PaO2 normal or baseline Patient is able to initiate an inspiratory effort Assess for home discharge ECMO Extracorporeal Membrane Oxygenation Version1.1 23 March 2020 Page 2 0f 4 COVID-19 Assessment for Mechanical Ventilation N.B. Dose and drug modifications are required for children below 12 years.
- 3. Definition of Suspected Cases Version1.1 23 March 2020 Page 3 0f 4 A. Any one of the epidemiological history with any of the clinical features. Epidemiological history: 1. History of travel to or residence in communities where cases reported within 14 days prior to the onset of the disease. 2. In contact with viral RNA positive people within 14 days prior to disease onset. 3. In contact with patients who have fever or respiratory symptoms from communities confirmed cases reported within 14 days before disease onset. B. All three clinical features. Clinical features: 1.Fever and/or respiratory symptoms. 2.Imaging characteristics. 3.The white blood cells was normal or decreased, with lymphocyte decreased. C. Severe Acute Respiratory Infection (SARI) with no other obvious cause. COVID-19 OR OR N.B. Healthcare providers exposed to suspected or confirmed COVID-19 case would follow the algorithm shown in MOHP guide booklet.
- 4. Treatment Protocol Revised By: Version1.1 23 March 2020 Page 4 0f 4 NAME Dr.Hossam Hosny Masoud Dr.Gehan Elassal Dr.Samy Zaki Dr.Amin Abdel Baky Dr.Hamdy Ibrahim Dr.Wagdy Amin Dr.Akram Abdelbary Dr.Ahmad Said Abdel Mohsen Dr.Mohamed Hassany Dr.Alaa Eid Dr.Noha Asem Mohamed Dr.Ehab Kamal Professor of Chest Diseases. Head of Pulmonary Hypertension Unit, Faculty of Medicine, Cairo University Professor of Pulmonary Medicine Ain Shams University Professor of Hepatogastroenterology and Infectious Diseases, Al Azhar University Consultant of Hepatoogy, Gastroenterology and Infectious Diseases. National Hepatology and Tropical Medicine Research Institute (NHTMRI), Cairo, Egypt General Director of Directorate of Fever Hospitals, MOHP Director General for Chest Diseases,MOHP Professor of critical care medicine, Cairo University Chairman elect of ELSO SWAAC chapter Lecturer of critical care medicine, Faculty of Medicine, Cairo University Fellow of Infectious Diseases and Endemic Hepatogastroentrology, National Hepatology and Tropical Medicine Research Institute Head of Preventive Medical Sector MOHP Minister's Advisor for Research and Health Development. Chairman of Research Ethics Committee MOHP. Lecturer of Public Health, Cairo University. Consultant Tropical Medicine Researcher of Tropical Medicine. Medical Division National Research Center. AFFILIATION COVID-19
