Photosyn
- 1. Chapter 7: Photosynthesis…using light to make fud 0
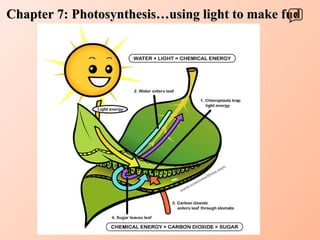
- 4. Carbon dioxide C 6 H 12 O 6 Photosynthesis H 2 O CO 2 O 2 Water + 6 6 Light energy Oxygen gas Glucose + 6
- 7. NPP: Examples Hmmm…what was NPP???
- 14. CO 2 O 2 Stoma Mesophyll Cell Vein Chloroplast Mesophyll Leaf Cross Section Leaf Outer and inner membranes Intermembrane space Granum Stroma Thylakoid space Thylakoid
- 15. CO 2 O 2 Stoma Mesophyll Cell Vein Chloroplast Mesophyll Leaf Cross Section Leaf
- 16. Chloroplast Outer and inner membranes Intermembrane space Granum Stroma Thylakoid space Thylakoid
- 24. H 2 O NADP + ADP P LIGHT REACTIONS (in thylakoids) Light Chloroplast
- 25. H 2 O ADP P LIGHT REACTIONS (in thylakoids) Light Chloroplast NADPH ATP O 2 Electrons NADP +
- 26. H 2 O ADP P LIGHT REACTIONS (in thylakoids) Light Chloroplast NADPH ATP O 2 Electrons CALVIN CYCLE (in stroma) Sugar CO 2 NADP +
- 29. Wavelength (nm) 10 –5 nm Increasing energy Visible light 650 nm 10 –3 nm 1 nm 10 3 nm 10 6 nm 1 m 10 3 m 380 400 500 600 700 750 Radio waves Micro- waves Infrared X-rays UV Gamma rays
- 32. Light Chloroplast Thylakoid Absorbed light Transmitted light Reflected light
- 34. Chlorophyll molecule Excited state Ground state Heat Photon Photon (fluorescence) e –
- 38. Reaction center complex e – Primary electron acceptor Light-harvesting complexes Photon Photosystem Transfer of energy Pigment molecules Pair of Chlorophyll a molecules Thylakoid membrane
- 39. NADPH Photosystem II e – Mill makes ATP Photon Photosystem I ATP e – e – e – e – e – e – Photon
- 40. Stroma O 2 H 2 O 1 2 H + NADP + NADPH Photon Photosystem II Electron transport chain Provides energy for synthesis of by chemiosmosis + 2 Primary acceptor 1 Thylakoid mem- brane P680 2 4 3 Thylakoid space e – e – 5 Primary acceptor P700 6 Photon Photosystem I ATP H + +
- 43. + O 2 H 2 O 1 2 H + NADP + H + NADPH + 2 H + H + H + H + H + H + H + H + H + H + H + H + H + H + Photosystem II Photosystem I Electron transport chain ATP synthase Light Light Stroma (low H + concentration) Chloroplast Thylakoid membrane Thylakoid space (high H + concentration) ADP + P ATP
- 44. + O 2 H 2 O 1 2 H + NADP + H + NADPH + 2 H + H + H + H + H + H + H + H + H + H + H + H + H + H + Photosystem II Photosystem I Electron transport chain ATP synthase Light Light Stroma (low H + concentration) Thylakoid space (high H + concentration) ADP + P ATP
- 46. CO 2 ATP NADPH Input C ALVIN CYCLE G3P Output:
- 49. NADP + NADPH ATP CO 2 + H 2 O ADP P Electron transport chains Thylakoid membranes Light Chloroplast O 2 C ALVIN C YCLE (in stroma) Sugars Photosystem II Photosystem I L IGHT R EACTIONS RuBP 3-PGA C ALVIN C YCLE Stroma G3P Cellular respiration Cellulose Starch Other organic compounds
- 50. Mesophyll cell CO 2 C ALVIN C YCLE CO 2 Bundle- sheath cell 3-C sugar C 4 plant 4-C compound CO 2 C ALVIN C YCLE CO 2 3-C sugar CAM plant 4-C compound Night Day
- 56. Atmosphere Sunlight Some heat energy escapes into space Radiant heat trapped by CO 2 and other gases
- 59. Southern tip of South America Antarctica
- 60. H 2 O ADP P Light reactions Light Chloroplast NADPH ATP O 2 Calvin cycle Sugar CO 2 NADP + Stroma Thylakoid membranes
- 61. Mitochondrion structure Intermembrane space Membrane Matrix a. H + Chloroplast structure b. c. d. e.
- 62. Photosynthesis includes both converts in which in which (b) (c) light-excited electrons of chlorophyll CO 2 is fixed to RuBP and then (h) reduce NADP + to using to produce sugar (G3P) (f) chemiosmosis (e) (g) by producing are passed down (d) and to chemical energy H 2 O is split (a)
Notas do Editor
- Photosynthesis nourishes almost the entire living world directly or indirectly. Almost all plants are autotrophs, meaning that they sustain themselves without eating anything derived from other living beings. Plants produce oxygen, a by-product of photosynthesis, that is used in respiration. The solar energy used in photosynthesis traveled 150 million kilometers from the sun to Earth to be converted into chemical energy. You may want to reintroduce the terms producers and consumers within the context of this chapter.
- A very interesting class of autotrophs are the autotrophic bacteria that use carbon dioxide to synthesize organic molecules without solar energy. Teaching Tips 1. When introducing the diverse ways that plants impact our lives, consider challenging your students to come up with a list of products made from plants that they come across on a regular basis. The collective lists from your students can be surprisingly long and might help to build up your catalog of examples. 2. The evolution of chloroplasts from photosynthetic prokaryotes living inside of eukaryotic cells is briefly noted in a reference to Module 4.16. If your students have not already read Chapter 4, consider discussing the evidence that suggests this endosymbiotic origin. 3. Some students might think that the term producers applies to the production of oxygen by plants. In turn, they might think that consumers are organisms that use oxygen (which would include all aerobic organisms). Extra care may be needed to clarify the definitions of these frequently used terms.
- There are about half a million chloroplasts per square millimeter of leaf surface. Chloroplast membranes are similar to mitochondrial membranes in that both are important in energy-harvesting duties of the cell. Teaching Tips 1. When introducing the diverse ways that plants impact our lives, consider challenging your students to come up with a list of products made from plants that they come across on a regular basis. The collective lists from your students can be surprisingly long and might help to build up your catalog of examples. 2. The evolution of chloroplasts from photosynthetic prokaryotes living inside of eukaryotic cells is briefly noted in a reference to Module 4.16. If your students have not already read Chapter 4, consider discussing the evidence that suggests this endosymbiotic origin. 3. Some students might think that the term producers applies to the production of oxygen by plants. In turn, they might think that consumers are organisms that use oxygen (which would include all aerobic organisms). Extra care may be needed to clarify the definitions of these frequently used terms.
- Figure 7.1A Forest plants.
- Figure 7.1B Wheat field.
- Figure 7.1C Kelp, a large algae.
- Figure 7.1D Micrograph of cyanobacteria (photosynthetic bacteria).
- Chlorophyll absorbs light energy and drives the synthesis of organic molecules. Teaching Tips 1. The authors note the analogous roles of the thylakoid space and the intermembrane space of a mitochondrion. Students might be encouraged to create a list of the similarities in structure and function of mitochondria and chloroplasts through these related chapters. 2. The living world contains many examples of adaptations to increase surface area. Some examples are the many folds of the inner mitochondrial membrane, the highly branched surfaces of fish gills and human lungs, and the highly branched system of capillaries in the tissues of our bodies. Consider relating this broad principle to the extensive folding of the thylakoid membranes.
- Leaves also use veins to export sugar to roots and other nonphotosynthetic parts of the plant. Teaching Tips 1. The authors note the analogous roles of the thylakoid space and the intermembrane space of a mitochondrion. Students might be encouraged to create a list of the similarities in structure and function of mitochondria and chloroplasts through these related chapters. 2. The living world contains many examples of adaptations to increase surface area. Some examples are the many folds of the inner mitochondrial membrane, the highly branched surfaces of fish gills and human lungs, and the highly branched system of capillaries in the tissues of our bodies. Consider relating this broad principle to the extensive folding of the thylakoid membranes.
- Chlorophyll resides in the thylakoid membranes. Teaching Tips 1. The authors note the analogous roles of the thylakoid space and the intermembrane space of a mitochondrion. Students might be encouraged to create a list of the similarities in structure and function of mitochondria and chloroplasts through these related chapters. 2. The living world contains many examples of adaptations to increase surface area. Some examples are the many folds of the inner mitochondrial membrane, the highly branched surfaces of fish gills and human lungs, and the highly branched system of capillaries in the tissues of our bodies. Consider relating this broad principle to the extensive folding of the thylakoid membranes.
- Figure 7.2 The location and structure of chloroplasts.
- Figure 7.2 The location and structure of chloroplasts.
- Figure 7.2 The location and structure of chloroplasts.
- C. B. van Niel of Stanford University hypothesized that plants split water into hydrogen and oxygen. His hypothesis was confirmed 20 years later. A significant result of photosynthesis is the extraction of hydrogen from water and its incorporation into sugar. Oxygen is a waste product of photosynthesis. The chloroplast is the site where water is split into hydrogen and oxygen. Student Misconceptions and Concerns 1. Students may not connect the growth in plant mass to the fixation of carbon during the Calvin cycle. It can be difficult for many students to appreciate that molecules in air can contribute significantly to the mass of plants. Teaching Tips 1. Many students do not realize that glucose is not the direct product of photosynthesis. The authors note that although glucose is shown as a product of photosynthesis, a three-carbon sugar is directly produced (G3P). A plant can use G3P to make many types of organic molecules, including glucose.
- Figure 7.3A Oxygen bubbles on the leaves of an aquatic plant.
- The simple sugar produced in photosynthesis is glucose, using a number of energy-releasing redox reactions. Teaching Tips 1. In our world, energy is frequently converted to a usable form in one place and used in another. For example, electricity is generated by power plants, transferred to our homes, and used to run computers, create light, and help us prepare foods. Consider relating this common energy transfer to the two-stage process of photosynthesis.
- In respiration, mitochondria harness chemical energy to synthesize ATP. In photosynthesis, the energy boost is provided by light and occurs in chloroplasts. Eventually, ATP is synthesized. Teaching Tips 1. In our world, energy is frequently converted to a usable form in one place and used in another. For example, electricity is generated by power plants, transferred to our homes, and used to run computers, create light, and help us prepare foods. Consider relating this common energy transfer to the two-stage process of photosynthesis.
- The sugar produced in photosynthesis is stored for later use or as raw material for biosynthesis of new plant material. Teaching Tips 1. In our world, energy is frequently converted to a usable form in one place and used in another. For example, electricity is generated by power plants, transferred to our homes, and used to run computers, create light, and help us prepare foods. Consider relating this common energy transfer to the two-stage process of photosynthesis.
- The two metabolic stages are the light reactions and the Calvin cycle. Student Misconceptions and Concerns 1. Students may understand the overall chemical relationships between photosynthesis and cellular respiration, but many struggle to understand the use of carbon dioxide in the Calvin cycle. Photosynthesis is much more than gas exchange. Teaching Tips 1. In our world, energy is frequently converted to a usable form in one place and used in another. For example, electricity is generated by power plants, transferred to our homes, and used to run computers, create light, and help us prepare foods. Consider relating this common energy transfer to the two-stage process of photosynthesis. 2. Figure 7.5 is an important visual organizer that notes the key structures and functions of the two stages of photosynthesis. This figure demonstrates where water and sunlight are used in the thylakoid membranes to generate oxygen, ATP, and NADPH. The second step, in the stroma, reveals the use of carbon dioxide, ATP, and NADPH to generate carbohydrates.
- The Calvin cycle was named for the Nobel laureate, Melvin Calvin, who traced the path of carbon in the cycle. Student Misconceptions and Concerns 1. Students may understand the overall chemical relationships between photosynthesis and cellular respiration, but many struggle to understand the use of carbon dioxide in the Calvin cycle. Photosynthesis is much more than gas exchange. Teaching Tips 1. In our world, energy is frequently converted to a usable form in one place and used in another. For example, electricity is generated by power plants, transferred to our homes, and used to run computers, create light, and help us prepare foods. Consider relating this common energy transfer to the two-stage process of photosynthesis. 2. Figure 7.5 is an important visual organizer that notes the key structures and functions of the two stages of photosynthesis. This figure demonstrates where water and sunlight are used in the thylakoid membranes to generate oxygen, ATP, and NADPH. The second step, in the stroma, reveals the use of carbon dioxide, ATP, and NADPH to generate carbohydrates.
- Figure 7.5 An overview of the two stages of photosynthesis that take place in a chloroplast. Figure 7.5 is an important visual organizer that notes the key structures and functions of the two stages of photosynthesis. This figure reminds students where water and sunlight are used in the thylakoid membranes to generate oxygen, ATP, and NADPH. The second step, in the stroma, reveals the use of carbon dioxide, ATP, and NADPH to generate carbohydrates.
- Figure 7.5 An overview of the two stages of photosynthesis that take place in a chloroplast. Figure 7.5 is an important visual organizer that notes the key structures and functions of the two stages of photosynthesis. This figure reminds students where water and sunlight are used in the thylakoid membranes to generate oxygen, ATP, and NADPH. The second step, in the stroma, reveals the use of carbon dioxide, ATP, and NADPH to generate carbohydrates.
- Figure 7.5 An overview of the two stages of photosynthesis that take place in a chloroplast. Figure 7.5 is an important visual organizer that notes the key structures and functions of the two stages of photosynthesis. This figure reminds students where water and sunlight are used in the thylakoid membranes to generate oxygen, ATP, and NADPH. The second step, in the stroma, reveals the use of carbon dioxide, ATP, and NADPH to generate carbohydrates.
- Student Misconceptions and Concerns 1. The authors note that electromagnetic energy travels through space in waves that are like ripples made by a pebble dropped in a pond. This wave imagery is helpful, but can confuse students when energy is also thought of as discrete packets called photons. The dual nature of light, which exhibits the properties of both waves and particles, may need to be discussed further, if students are to do more than just accept the definitions. 2. The authors note that sunlight is a type of radiation. Many students think of radiation as a result of radioactive decay, a serious threat to health. The diverse types of radiation and the varying energy associated with each might need to be explained. 3. Even at the college level, students struggle to understand why we perceive certain colors. The authors discuss the specific absorption and reflection of certain wavelengths of light, noting which colors are absorbed and which are reflected (and thus available for our eyes to detect). Consider spending time to make sure that your students understand how photosynthetic pigments absorb and reflect certain wavelengths. Teaching Tips 1. Consider bringing a prism to class and demonstrating the spectrum of light. Depending on what you have available, it can be a dramatic reinforcement.
- Figure 7.6A The electromagnetic spectrum and the wavelengths of visible light. (A wavelength of 650 nm is illustrated.)
- Each type of pigment absorbs certain wavelengths of light because it is able to absorb the specific amount of energy in those photons. Student Misconceptions and Concerns 1. The authors note that electromagnetic energy travels through space in waves that are like ripples made by a pebble dropped in a pond. This wave imagery is helpful, but can confuse students when energy is also thought of as discrete packets called photons. The dual nature of light, which exhibits the properties of both waves and particles, may need to be discussed further, if students are to do more than just accept the definitions. 2. The authors note that sunlight is a type of radiation. Many students think of radiation as a result of radioactive decay, a serious threat to health. The diverse types of radiation and the varying energy associated with each might need to be explained. 3. Even at the college level, students struggle to understand why we perceive certain colors. The authors discuss the specific absorption and reflection of certain wavelengths of light, noting which colors are absorbed and which are reflected (and thus available for our eyes to detect). Consider spending time to make sure that your students understand how photosynthetic pigments absorb and reflect certain wavelengths. Teaching Tips 1. Consider bringing a prism to class and demonstrating the spectrum of light. Depending on what you have available, it can be a dramatic reinforcement.
- Figure 7.6B The interaction of light with a chloroplast.
- The colors of fall foliage in certain parts of the world are due partly to the yellow-orange hues of longer lasting carotenoids that show through once the green chlorophyll breaks down. Student Misconceptions and Concerns 1. The authors note that electromagnetic energy travels through space in waves that are like ripples made by a pebble dropped in a pond. This wave imagery is helpful, but can confuse students when energy is also thought of as discrete packets called photons. The dual nature of light, which exhibits the properties of both waves and particles, may need to be discussed further, if students are to do more than just accept the definitions. 2. The authors note that sunlight is a type of radiation. Many students think of radiation as a result of radioactive decay, a serious threat to health. The diverse types of radiation and the varying energy associated with each might need to be explained. 3. Even at the college level, students struggle to understand why we perceive certain colors. The authors discuss the specific absorption and reflection of certain wavelengths of light, noting which colors are absorbed and which are reflected (and thus available for our eyes to detect). Consider spending time to make sure that your students understand how photosynthetic pigments absorb and reflect certain wavelengths. Teaching Tips 1. Consider bringing a prism to class and demonstrating the spectrum of light. Depending on what you have available, it can be a dramatic reinforcement.
- Figure 7.7A Light-excited chlorophyll molecule that releases a photon of red light.
- Because of their functions, you can think of photosystems as light-gathering antennae. Student Misconceptions and Concerns 1. Even at the college level, students struggle to understand why we perceive certain colors. The authors discuss the specific absorption and reflection of certain wavelengths of light, noting which colors are absorbed and which are reflected (and thus available for our eyes to detect). Consider spending time to make sure that your students understand how photosynthetic pigments absorb and reflect certain wavelengths. Teaching Tips 1. The authors discuss a phenomenon that most students have noticed: dark surfaces heat up faster in the sun than do lighter-colored surfaces. This is an opportunity to demonstrate to your students the various depths of scientific explanations and help them appreciate their own educational progress. In elementary school, they might have learned that the sun heats darker surfaces faster than lighter surfaces. In high school, they may have learned about light energy and the fact that dark surfaces absorb more of this energy than lighter surfaces. Now, in college, they are learning that at the atomic level, darker surfaces absorb the energy of more photons, exciting more electrons, which then fall back to a lower state, releasing more heat.
- Student Misconceptions and Concerns 1. Even at the college level, students struggle to understand why we perceive certain colors. The authors discuss the specific absorption and reflection of certain wavelengths of light, noting which colors are absorbed and which are reflected (and thus available for our eyes to detect). Consider spending time to make sure that your students understand how photosynthetic pigments absorb and reflect certain wavelengths. Teaching Tips 1. The authors discuss a phenomenon that most students have noticed: dark surfaces heat up faster in the sun than do lighter-colored surfaces. This is an opportunity to demonstrate to your students the various depths of scientific explanations and help them appreciate their own educational progress. In elementary school, they might have learned that the sun heats darker surfaces faster than lighter surfaces. In high school, they may have learned about light energy and the fact that dark surfaces absorb more of this energy than lighter surfaces. Now, in college, they are learning that at the atomic level, darker surfaces absorb the energy of more photons, exciting more electrons, which then fall back to a lower state, releasing more heat.
- The photosystems were named in order of their discovery, not in order of their function. Student Misconceptions and Concerns 1. Even at the college level, students struggle to understand why we perceive certain colors. The authors discuss the specific absorption and reflection of certain wavelengths of light, noting which colors are absorbed and which are reflected (and thus available for our eyes to detect). Consider spending time to make sure that your students understand how photosynthetic pigments absorb and reflect certain wavelengths. Teaching Tips 1. The authors discuss a phenomenon that most students have noticed: dark surfaces heat up faster in the sun than do lighter-colored surfaces. This is an opportunity to demonstrate to your students the various depths of scientific explanations and help them appreciate their own educational progress. In elementary school, they might have learned that the sun heats darker surfaces faster than lighter surfaces. In high school, they may have learned about light energy and the fact that dark surfaces absorb more of this energy than lighter surfaces. Now, in college, they are learning that at the atomic level, darker surfaces absorb the energy of more photons, exciting more electrons, which then fall back to a lower state, releasing more heat.
- Figure 7.7B Light-excited chlorophyll embedded in a photosystem: Its electron is transferred to a primary electron acceptor before it returns to ground state.
- Figure 7.8B A mechanical analogy of the light reactions. Although Figure 7.8B can be very effective, students might need to be carefully led through the analogy to understand precisely what is represented.
- Figure 7.8A Electron flow in the light reactions of photosynthesis: Both photosystems and the electron transport chain that connects them are located in the thylakoid membrane. The energy from light drives electrons from water to NADPH.
- The gradient is produced as the electron transport chain passes electrons down the chain. Teaching Tips 1. Module 7.9 notes the similarities between oxidative phosphorylation in mitochondria and photophosphorylation in chloroplasts. If your students have not already read or discussed chemiosmosis in mitochondria, consider assigning Modules 6.6 and 6.10 to show the similarities of these processes. (As noted in Module 7.2, the thylakoid space is analogous to the intermembrane space of mitochondria.)
- Students should realize that electrons flowing between the two photosystems do not end up at a low energy level in water as they do in respiration; instead they are stored at a high state of potential energy in NADPH. Teaching Tips 1. Module 7.9 notes the similarities between oxidative phosphorylation in mitochondria and photophosphorylation in chloroplasts. If your students have not already read or discussed chemiosmosis in mitochondria, consider assigning Modules 6.6 and 6.10 to show the similarities of these processes. (As noted in Module 7.2, the thylakoid space is analogous to the intermembrane space of mitochondria.)
- Figure 7.9 The production of ATP by chemiosmosis in photosynthesis: The small diagram on the upper left illustrates the location of the components of the light reactions in a thylakoid membrane. Numerous copies of these components are present in each thylakoid.
- Figure 7.9 The production of ATP by chemiosmosis in photosynthesis.
- Figure 7.10A An overview of the Calvin cycle.
- Although photosynthesizers produce sugar for self-consumption, their sugar is a source for virtually all other organisms on Earth. Student Misconceptions and Concerns 1. Some students do not realize that plant cells also have mitochondria. Instead, they assume that the chloroplasts are sufficient for the plant cell’s needs. As noted in the text, nearly 50% of the carbohydrates produced by plant cells are used for cellular respiration (involving mitochondria). Teaching Tips 1. Challenge students to explain how the energy in beef is ultimately derived from the sun. 2. The authors note that G3P is also used to produce cellulose, the most abundant compound on Earth. Each year, plants produce about 100 billion tons of cellulose!
- Figure 7.11 A summary of the chemical processes of photosynthesis.
- Figure 7.12 Comparison of photosynthesis in C 4 and CAM plants: In both pathways, CO 2 is first incorporated into a four-carbon compound, which then provides CO 2 to the Calvin cycle.
- Student Misconceptions and Concerns 1. Students may confuse global warming with the breakdown of the ozone layer. Be prepared to explain both phenomena and the impact of human activities. 2. Students often do not fully understand how the burning of fossil fuels contributes to global warming. They might wonder, “How does the burning of fossil fuels differ from the burning of ethanol produced from crops?” Students might not realize that the carbon in fossil fuels was removed from the atmosphere hundreds of millions of years ago, while the carbon in crops was removed much more recently, when the crops were grown. Teaching Tips 1. Some students might better relate the greenhouse effect to what happens inside their closed car on a sunny day. The glass in our automobiles functions just like the glass of a greenhouse, trapping heat inside our car. This can be an advantage during the winter but is usually not welcome on a hot summer day!
- Student Misconceptions and Concerns 1. Students may confuse global warming with the breakdown of the ozone layer. Be prepared to explain both phenomena and the impact of human activities. 2. Students often do not fully understand how the burning of fossil fuels contributes to global warming. They might wonder, “How does the burning of fossil fuels differ from the burning of ethanol produced from crops?” Students might not realize that the carbon in fossil fuels was removed from the atmosphere hundreds of millions of years ago, while the carbon in crops was removed much more recently, when the crops were grown. Teaching Tips 1. Some students might better relate the greenhouse effect to what happens inside their closed car on a sunny day. The glass in our automobiles functions just like the glass of a greenhouse, trapping heat inside our car. This can be an advantage during the winter but is usually not welcome on a hot summer day!
- Student Misconceptions and Concerns 1. Students may confuse global warming with the breakdown of the ozone layer. Be prepared to explain both phenomena and the impact of human activities. 2. Students often do not fully understand how the burning of fossil fuels contributes to global warming. They might wonder, “How does the burning of fossil fuels differ from the burning of ethanol produced from crops?” Students might not realize that the carbon in fossil fuels was removed from the atmosphere hundreds of millions of years ago, while the carbon in crops was removed much more recently, when the crops were grown. Teaching Tips 1. Some students might better relate the greenhouse effect to what happens inside their closed car on a sunny day. The glass in our automobiles functions just like the glass of a greenhouse, trapping heat inside our car. This can be an advantage during the winter but is usually not welcome on a hot summer day!
- Figure 7.13A Plants growing in a greenhouse.
- Figure 7.13B CO 2 in the atmosphere and global warming.
- Student Misconceptions and Concerns 1. Students may confuse global warming with the breakdown of the ozone layer. Be prepared to explain both phenomena and the impact of human activities. Teaching Tips 1. Consider an analogy between the ozone layer and sunscreen applied to the skin. The thinning of the ozone layer is like putting on less and less sunscreen. In both situations, more harmful UV light penetrates the layers and causes damage. 2. Frustration can overwhelm concerned students alarmed by the many problems addressed in this chapter. One way to address this is to provide meaningful ways for students to respond to this information (for example, changes in personal choices and voting). The Earth Day Network, www.earthday.net, is just one of many Internet sites devoted to positive action.
- Figure 7.14A Mario Molina.
- Figure 7.14B The ozone hole in the Southern Hemisphere, spring 2006.