1 25 What Is P H
•
0 gostou•663 visualizações
Denunciar
Compartilhar
Denunciar
Compartilhar
Baixar para ler offline
Recomendados
Recomendados
Mais conteúdo relacionado
Mais procurados
Mais procurados (19)
Concentration of a solution ||5. Acids, Bases and Salts || 

Concentration of a solution ||5. Acids, Bases and Salts ||
IB Chemistry on Titration Curves between Acids and Bases

IB Chemistry on Titration Curves between Acids and Bases
Semelhante a 1 25 What Is P H
Semelhante a 1 25 What Is P H (20)
Gifted Students - Acids & Bases - The Gifted Child - Giftedness Enrichment Wo...

Gifted Students - Acids & Bases - The Gifted Child - Giftedness Enrichment Wo...
Mais de mrheffner
Mais de mrheffner (20)
Último
☑️░ 9630942363 ░ CALL GIRLS ░ VIP ░ ESCORT ░ SERVICES ░ AGENCY ░
9630942363 THE GENUINE ESCORT AGENCY VIP LUXURY CALL GIRLS
HIGH CLASS MODELS CALL GIRLS GENUINE ESCORT BOOK
BOOK APPOINTMENT - 9630942363 THE GENUINE ESCORT AGENCY
BEST VIP CALL GIRLS & ESCORTS SERVICE 9630942363 VIP CALL GIRLS ALL TYPE WOMEN AVAILABLE
INCALL & OUTCALL BOTH AVAILABLE BOOK NOW
9630942363 VIP GENUINE INDEPENDENT ESCORT AGENCY
VIP PRIVATE AUNTIES
BEAUTIFUL LOOKING HOT AND SEXT GIRLS AND PARTY TYPE GIRLS YOU WANT SERVICE THEN CALL THIS NUMBER 9630942363
ROOM ALSO PROVIDE HOME & HOTELS SERVICE
FULL SAFE AND SECURE WORK
WITHOUT CONDOMS, ORAL, SUCKING, LIP TO LIP, ANAL, BACK SHOTS, SEX 69, WITHOUT BLOWJOB AND MUCH MORE
FOR BOOKING
9630942363Call Girls Vasai Virar Just Call 9630942363 Top Class Call Girl Service Avail...

Call Girls Vasai Virar Just Call 9630942363 Top Class Call Girl Service Avail...GENUINE ESCORT AGENCY
🌹Attapur⬅️ Vip Call Girls Hyderabad 📱9352852248 Book Well Trand Call Girls In Hyderabad Escorts Service
Escorts Service Available
Whatsapp Chaya ☎️ : [+91-9352852248 ]
Escorts Service Hyderabad are always ready to make their clients happy. Their exotic looks and sexy personalities are sure to turn heads. You can enjoy with them, including massages and erotic encounters.#P12Our area Escorts are young and sexy, so you can expect to have an exotic time with them. They are trained to satiate your naughty nerves and they can handle anything that you want. They are also intelligent, so they know how to make you feel comfortable and relaxed
SERVICE ✅ ❣️
⭐➡️HOT & SEXY MODELS // COLLEGE GIRLS HOUSE WIFE RUSSIAN , AIR HOSTES ,VIP MODELS .
AVAILABLE FOR COMPLETE ENJOYMENT WITH HIGH PROFILE INDIAN MODEL AVAILABLE HOTEL & HOME
★ SAFE AND SECURE HIGH CLASS SERVICE AFFORDABLE RATE
★
SATISFACTION,UNLIMITED ENJOYMENT.
★ All Meetings are confidential and no information is provided to any one at any cost.
★ EXCLUSIVE PROFILes Are Safe and Consensual with Most Limits Respected
★ Service Available In: - HOME & HOTEL Star Hotel Service .In Call & Out call
SeRvIcEs :
★ A-Level (star escort)
★ Strip-tease
★ BBBJ (Bareback Blowjob)Receive advanced sexual techniques in different mode make their life more pleasurable.
★ Spending time in hotel rooms
★ BJ (Blowjob Without a Condom)
★ Completion (Oral to completion)
★ Covered (Covered blowjob Without condom
★ANAL SERVICES.
🌹Attapur⬅️ Vip Call Girls Hyderabad 📱9352852248 Book Well Trand Call Girls In...

🌹Attapur⬅️ Vip Call Girls Hyderabad 📱9352852248 Book Well Trand Call Girls In...Call Girls In Delhi Whatsup 9873940964 Enjoy Unlimited Pleasure
9630942363 THE GENUINE ESCORT AGENCY VIP LUXURY CALL GIRLS
HIGH CLASS MODELS CALL GIRLS GENUINE ESCORT BOOK
BOOK APPOINTMENT - 9630942363 THE GENUINE ESCORT AGENCY
BEST VIP CALL GIRLS & ESCORTS SERVICE 9630942363 VIP CALL GIRLS ALL TYPE WOMEN AVAILABLE
INCALL & OUTCALL BOTH AVAILABLE BOOK NOW
9630942363 VIP GENUINE INDEPENDENT ESCORT AGENCY
VIP PRIVATE AUNTIES
BEAUTIFUL LOOKING HOT AND SEXT GIRLS AND PARTY TYPE GIRLS YOU WANT SERVICE THEN CALL THIS NUMBER 9630942363
ROOM ALSO PROVIDE HOME & HOTELS SERVICE
FULL SAFE AND SECURE WORK
WITHOUT CONDOMS, ORAL, SUCKING, LIP TO LIP, ANAL, BACK SHOTS, SEX 69, WITHOUT BLOWJOB AND MUCH MORE
FOR BOOKING
9630942363Call Girls Ahmedabad Just Call 9630942363 Top Class Call Girl Service Available

Call Girls Ahmedabad Just Call 9630942363 Top Class Call Girl Service AvailableGENUINE ESCORT AGENCY
☑️░ 9630942363 ░ CALL GIRLS ░ VIP ░ ESCORT ░ SERVICES ░ AGENCY ░
9630942363 THE GENUINE ESCORT AGENCY VIP LUXURY CALL GIRLS
HIGH CLASS MODELS CALL GIRLS GENUINE ESCORT BOOK
BOOK APPOINTMENT - 9630942363 THE GENUINE ESCORT AGENCY
BEST VIP CALL GIRLS & ESCORTS SERVICE 9630942363 VIP CALL GIRLS ALL TYPE WOMEN AVAILABLE
INCALL & OUTCALL BOTH AVAILABLE BOOK NOW
9630942363 VIP GENUINE INDEPENDENT ESCORT AGENCY
VIP PRIVATE AUNTIES
BEAUTIFUL LOOKING HOT AND SEXT GIRLS AND PARTY TYPE GIRLS YOU WANT SERVICE THEN CALL THIS NUMBER 9630942363
ROOM ALSO PROVIDE HOME & HOTELS SERVICE
FULL SAFE AND SECURE WORK
WITHOUT CONDOMS, ORAL, SUCKING, LIP TO LIP, ANAL, BACK SHOTS, SEX 69, WITHOUT BLOWJOB AND MUCH MORE
FOR BOOKING
9630942363Trichy Call Girls Book Now 9630942363 Top Class Trichy Escort Service Available

Trichy Call Girls Book Now 9630942363 Top Class Trichy Escort Service AvailableGENUINE ESCORT AGENCY
☑️░ 9630942363 ░ CALL GIRLS ░ VIP ░ ESCORT ░ SERVICES ░ AGENCY ░
9630942363 THE GENUINE ESCORT AGENCY VIP LUXURY CALL GIRLS
HIGH CLASS MODELS CALL GIRLS GENUINE ESCORT BOOK
BOOK APPOINTMENT - 9630942363 THE GENUINE ESCORT AGENCY
BEST VIP CALL GIRLS & ESCORTS SERVICE 9630942363 VIP CALL GIRLS ALL TYPE WOMEN AVAILABLE
INCALL & OUTCALL BOTH AVAILABLE BOOK NOW
9630942363 VIP GENUINE INDEPENDENT ESCORT AGENCY
VIP PRIVATE AUNTIES
BEAUTIFUL LOOKING HOT AND SEXT GIRLS AND PARTY TYPE GIRLS YOU WANT SERVICE THEN CALL THIS NUMBER 9630942363
ROOM ALSO PROVIDE HOME & HOTELS SERVICE
FULL SAFE AND SECURE WORK
WITHOUT CONDOMS, ORAL, SUCKING, LIP TO LIP, ANAL, BACK SHOTS, SEX 69, WITHOUT BLOWJOB AND MUCH MORE
FOR BOOKING
9630942363Pondicherry Call Girls Book Now 9630942363 Top Class Pondicherry Escort Servi...

Pondicherry Call Girls Book Now 9630942363 Top Class Pondicherry Escort Servi...GENUINE ESCORT AGENCY
Último (20)
8980367676 Call Girls In Ahmedabad Escort Service Available 24×7 In Ahmedabad

8980367676 Call Girls In Ahmedabad Escort Service Available 24×7 In Ahmedabad
Call Girls Vasai Virar Just Call 9630942363 Top Class Call Girl Service Avail...

Call Girls Vasai Virar Just Call 9630942363 Top Class Call Girl Service Avail...
The Most Attractive Hyderabad Call Girls Kothapet 𖠋 9332606886 𖠋 Will You Mis...

The Most Attractive Hyderabad Call Girls Kothapet 𖠋 9332606886 𖠋 Will You Mis...
Call Girls Vadodara Just Call 8617370543 Top Class Call Girl Service Available

Call Girls Vadodara Just Call 8617370543 Top Class Call Girl Service Available
Call Girls Guntur Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Guntur Just Call 8250077686 Top Class Call Girl Service Available
🌹Attapur⬅️ Vip Call Girls Hyderabad 📱9352852248 Book Well Trand Call Girls In...

🌹Attapur⬅️ Vip Call Girls Hyderabad 📱9352852248 Book Well Trand Call Girls In...
Best Rate (Patna ) Call Girls Patna ⟟ 8617370543 ⟟ High Class Call Girl In 5 ...

Best Rate (Patna ) Call Girls Patna ⟟ 8617370543 ⟟ High Class Call Girl In 5 ...
Top Rated Hyderabad Call Girls Chintal ⟟ 9332606886 ⟟ Call Me For Genuine Se...

Top Rated Hyderabad Call Girls Chintal ⟟ 9332606886 ⟟ Call Me For Genuine Se...
Call Girls Ahmedabad Just Call 9630942363 Top Class Call Girl Service Available

Call Girls Ahmedabad Just Call 9630942363 Top Class Call Girl Service Available
Call Girls Service Jaipur {9521753030} ❤️VVIP RIDDHI Call Girl in Jaipur Raja...

Call Girls Service Jaipur {9521753030} ❤️VVIP RIDDHI Call Girl in Jaipur Raja...
Top Rated Bangalore Call Girls Ramamurthy Nagar ⟟ 9332606886 ⟟ Call Me For G...

Top Rated Bangalore Call Girls Ramamurthy Nagar ⟟ 9332606886 ⟟ Call Me For G...
Call Girls Shimla Just Call 8617370543 Top Class Call Girl Service Available

Call Girls Shimla Just Call 8617370543 Top Class Call Girl Service Available
Top Rated Bangalore Call Girls Majestic ⟟ 9332606886 ⟟ Call Me For Genuine S...

Top Rated Bangalore Call Girls Majestic ⟟ 9332606886 ⟟ Call Me For Genuine S...
Call Girls Tirupati Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Tirupati Just Call 8250077686 Top Class Call Girl Service Available
Trichy Call Girls Book Now 9630942363 Top Class Trichy Escort Service Available

Trichy Call Girls Book Now 9630942363 Top Class Trichy Escort Service Available
All Time Service Available Call Girls Marine Drive 📳 9820252231 For 18+ VIP C...

All Time Service Available Call Girls Marine Drive 📳 9820252231 For 18+ VIP C...
Call Girls Rishikesh Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Rishikesh Just Call 8250077686 Top Class Call Girl Service Available
VIP Hyderabad Call Girls Bahadurpally 7877925207 ₹5000 To 25K With AC Room 💚😋

VIP Hyderabad Call Girls Bahadurpally 7877925207 ₹5000 To 25K With AC Room 💚😋
Independent Call Girls In Jaipur { 8445551418 } ✔ ANIKA MEHTA ✔ Get High Prof...

Independent Call Girls In Jaipur { 8445551418 } ✔ ANIKA MEHTA ✔ Get High Prof...
Pondicherry Call Girls Book Now 9630942363 Top Class Pondicherry Escort Servi...

Pondicherry Call Girls Book Now 9630942363 Top Class Pondicherry Escort Servi...
1 25 What Is P H
- 1. Launch: 1/25 Grab your binder and immediately take a seat! Place launch paper and Friday’s practice questions on your desk. Friday’s Quiz Data: 94%! Today’s Objective: I can use the pH scale to characterize acid and base solutions.
- 2. Launch: 1/25 Grab your binder and immediately take a seat! Place launch paper and Friday’s practice questions on your desk. Friday’s Quiz Data: 89%! Today’s Objective: I can use the pH scale to characterize acid and base solutions.
- 3. Launch: 1/25 Grab your binder and immediately take a seat! Place launch paper and Friday’s practice questions on your desk. Friday’s Quiz Data: 95%! Today’s Objective: I can use the pH scale to characterize acid and base solutions.
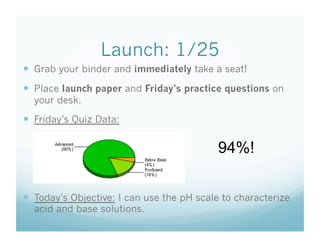
- 4. Launch: 1/25 Grab your binder and immediately take a seat! Place launch paper and Friday’s practice questions on your desk. Friday’s Quiz Data: 94%! Today’s Objective: I can use the pH scale to characterize acid and base solutions.
- 5. Launch 1/25 1. Draw a picture of • HCl (a strong acid) dissolved in water • HF (a weak acid) dissolved in water
- 6. Launch 1/25 2. In a complete sentence, describe the difference between strong and weak acids. (Hint: use the word “dissociation”)
- 7. Launch 1/25 3. In the equation, HSO4- + H2O H2SO4 + OH-, which reactant molecule acts as the base? a. HSO4- b. H2O c. H2SO4 d. OH-
- 8. Announcements Unit #6 Exam this Thursday! Ask questions Work with purpose Class rules I am focusing on this week: interfering with your own or someone else’s learning leaving seat without permission
- 9. January Calendar Exam
- 10. What is pH? Mr. Heffner 1/25/10
- 11. Review: What are strong acids/bases? strong acid + strong base = water + a salt neutralize each other (pH 7) an ionic compound HI + KOH ? H+ + l- + K+ + OH- ? 2O + KI H a salt water
- 12. What is pH? pH is… the concentration of H+ ions in a solution pH = - log( H+ ) the molarity function on H+ ions of a calculator (mol/L)
- 13. What is pH? The pH scale is not linear it is logarithmic pH = - log( H+ ) Meaning… 1 pH unit = 10 times the acidity stomach acid (pH 1) is 10X more acidic than lemon juice (pH 2)
- 14. What is pH? For each 1 pH unit difference, add a “0” stomach acid (pH 1) is 10X more acidic than 1000X more acidic than 100X more acidic than lemon soda (pH 3) vinegar (pH 2) juice 4) Three“0”s b/c pH Two “0”s b/c pH difference = 2 difference = 3 This is because the pH scale is logarithmic!
- 15. Whiteboards Work in pairs Trade-off marker every question Partner without the marker should be checking work at each step! You have 60 seconds to answer the question Which is more more acidic/basic? How much more acidic/basic? Lift board only when prompted
- 16. Lemon juice (pH 2) Soda (pH 4) Which is more acidic and by how much?
- 17. Ammonia (pH 10) Water (pH 7) Which is more basic and by how much?
- 18. Rainwater (pH 5) 10M HCl (pH 0) Which is the stronger acid and by how much?
- 19. Bleach (pH 12) Baking soda (pH 9) Which is the stronger base and by how much?
- 20. Tomato Lemon Which is the stronger acid and by how much?
- 21. Banana Soap Which is the stronger base and by how much?
- 22. Rainwater Pure water Which is the stronger acid and by how much?
- 23. Drain cleaner Egg whites Which is the stronger base and by how much?
- 24. Practice Questions Practice question worksheet
- 25. Binder Re-label your tabs: 1. Launches 2. Notes 3. Practice Questions 4. Handouts/Labs 5. Grades
- 26. Exit Slip 1. pH is a measure of a. the taste of an acid or a base. b. the ability of a solute to dissolve in solution. c. the concentration of H+ ions in solution. d. the strength of an ion in solution.
- 27. Exit Slip 2. The pH scale is a. logarithmic, meaning that 1 pH unit equals ten times the acidity/basicity b. logarithmic, meaning that 1 pH unit equals one times the acidity/basicity c. linear, meaning that 1 pH unit equals ten times the acidity/basicity d. linear, meaning that 1 pH unit equals one times the acidity/basicity
- 28. Exit Slip 3. Of the four different laboratory solutions below, the solution with the highest acidity has a pH of a. 11. b. 7. c. 5. d. 3.
- 29. Exit Slip 4. How many times more basic is drain cleaner (pH 12) than baking soda (pH 9)? a. 3 b. 30 c. 100 d. 1000
- 30. Exit Slip 5. How many times more acidic is stomach acid than rainwater? a. 4 b. 10000 c. 40000 d. 100000
- 31. Homework Finish practice questions