Mais conteúdo relacionado
Semelhante a Pdf tabla-de-entalpias-energia-libre-de-gibbs-y-entropia compress (20)
Pdf tabla-de-entalpias-energia-libre-de-gibbs-y-entropia compress
- 1. tu
tu
u
u
■
■u
ui
i
C
C
u
u
C
C
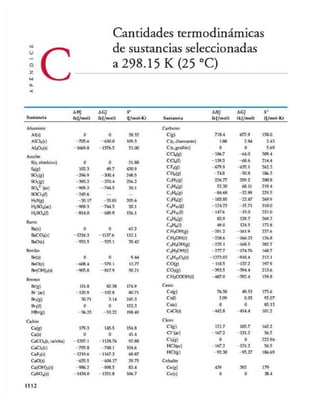
Cantidades termodinámicas
Cantidades termodinámicas
de sustancias seleccionadas
de sustancias seleccionadas
a 298.15 K (25 °C)
a 298.15 K (25 °C)
Sustancia
Sustancia
A
AH
H
°
°f
f
(kj/mol)
(kj/mol)
A
AG
GJ
J
(kj/mol)
(kj/mol)
S°
S°
(J/mol-K)
(J/mol-K) Sustanda
Sustanda
a
a h
h ;
;
(kj/mol)
(kj/mol)
A
AG
GJ
J
(kj/mol)
(kj/mol)
S°
S°
(J/mol-K)
(J/mol-K)
A
Al
lu
um
mi
in
ni
io
o C
Ca
ar
rb
bo
on
no
o
Al(s)
Al(s) 0
0 0
0 28.32
28.32 c
c (g)
(g) 7
71
18
8.
.4
4 6
67
72
2.
.9 1
9 15
58
8.
.0
0
A1C1
A1C13
3(s)
(s) -
-7
70
05
5.
.6
6 -
-6
63
30
0.
.0
0 1
10
09
9.
.3
3 C(s,
C(s, diamante)
diamante) 1.88
1.88 2
2.
.8
84
4 2
2.
.4
43
3
A
AI
I2
2Chis)
Chis) -
-1
16
66
69
9.
.8 - 1
8 - 15
57
76
6.
.5
5 5
51
1.
.0
00
0 C(s,
C(s, grafito)
grafito) 0
0 0
0 5.69
5.69
Azufre
Azufre c
ce
ek(g)
k(g) -
-1
10
06
6.
.7
7 -
-6
64
4.
.0
0 3
30
09
9.
.4
4
S(s, rómbico)
S(s, rómbico) 0
0 0
0 31.88
31.88 CCk(l)
CCk(l) -
-1
13
39
93
3 -
- 68.6
68.6 214.4
214.4
Ss
Ss(g)
(g) 1
10
02
2.
.3
3 4
49
9.
.7
7 4
43
30
0.
.9
9 C
CF
F4
4(g)
(g) -
-6
67
79
9.
.9
9 -
-6
63
35
5.
.1
1 2
26
62
2.
.3
3
SÜ
SÜ2
2(g)
(g) -
-2
29
96
6.
.9
9 -
-3
30
00
0.
.4
4 2
24
48
8.
.5
5 CH»
CH»(g)
(g) —
—7
74
4.
.8
8 -
-5
50
0.
.8
8 1
18
86
6.
.3
3
S
S03
036
6)
) -
-3
39
95
5.
.2
2 -
-3
37
70
0.
.4
4 2
25
56
6.
.2
2 C2H
C2H2
2(
(g
g)
) 2
22
26
6.
.7
77
7 2
20
09
9.
.2
2 200.8
200.8
SO
SO42
42
_(flc)
_(flc) -
-9
90
09
9.
.3
3 -
-7
74
44
4.
.5
5 20.1
20.1 C
C2
2H
H4
4{g)
{g) 52.30
52.30 68.11
68.11 219.4
219.4
S
SO
OC
Cl
l2
2(
(/
/)
) -
-2
24
45
5.
.6
6 —
— —
— C
Cí
í H<&)
H<&) -
-8
84
4.
.6
68
8 -
-3
32
2.
.8
89 2
9 22
29
9.
.5
5
H
H2
2S(£)
S(£) -
-2
20
0.
.1
17
7 -
-3
33
3.
.0
01
1 2
20
05
5.
.6
6 C
C3
3H
H8
8(
(g
g)
) -
-1
10
03
3.
.8
85
5 -
-2
23
3.
.4
47 2
7 26
69
9.
.9
9
H
H2
2S 0
S 0 4
4(
(«
«c
c)
) -
-9
90
09
9.
.3
3 -
-7
74
44
4.
.5
5 20.1
20.1 C
C4H
4HM
Mfe)
fe) -
-1
12
24
4.
.7
73
3 -
-1
15
5.
.7
71
1 3
31
10
0.
.0
0
h
h 2
2s o
s o 4(/)
4(/) -
-8
81
14
4.
.0
0 -
-6
68
89
9.
.9
9 1
15
56
6.
.1
1 c
c 4
4h
h 10(0
10(0 -
-1
14
47
7.
.6
6 -
-1
15
5.
.0
0 2
23
31
1.
.0
0
Bario
Bario
CéH
CéH6
6
(g)
(g) 8
82
2.
.9
9 1
12
29
9.
.7 2
7 26
69
9.
.2
2
C
C ^
^ O
O 4
49
9.
.0
0 1
12
24
4.
.5 1
5 17
72
2.
.8
8
Ba(s)
Ba(s) 0
0 0
0 63.2
63.2
CH
CH3
3OH
OH(g)
(g) -
- 201.2
201.2 -
-1
16
61
1.
.9
9 2
23
37
7.
.6
6
BaCO^s)
BaCO^s) -1
-121
216.
6.3 -11
3 -1137
37.
.6
6 112.1
112.1
C
CH
H3
3O
OH
H(
(/
/)
) -
-2
23
38
8.
.6
6 -
-1
16
66
6.
.2
23
3 1
12
26
6.
.8
8
BaO(s)
BaO(s) -
-5
55
53
3.
.5
5 -
-5
52
25
5.
.1
1 7
70
0.
.4
42
2
C2HsOH
C2HsOH(g)
(g) -
-2
23
35
5.
.1
1 -
-1
16
68
8.
.5
5 2
28
82
2.
.7
7
B
Be
er
ri
il
li
io
o C
C2
2H
Hs
sO
OH
H(
(/
/)
) -
-2
27
77
7.
.7
7 -
-1
17
74
4.
.7
76
6 1
16
60
0.
.7
7
Be(s)
Be(s) 0
0 0
0 9.44
9.44 C
C ¿
¿ ín
ín 0
0 6
6(s)
(s) -
-12
1273
73.
.02
02 -9
-910
10.4
.4 212.1
212.1
BeO(s)
BeO(s) -
-6
60
08
8.
.4
4 -
-5
57
79
9.
.1
1 1
13
3.
.7
77
7 CO
CO (g)
(g) -
-1
11
10
0.
.5
5 -
-1
13
37
7.
.2
2 1
19
97
7.
.9
9
Be(OH)
Be(OH)2
2(s)
(s) -
-9
90
05
5.
.8
8 -
-8
81
17
7.
.9
9 5
50
0.
.2
21
1 C
C0
02
2(
(g
g)
) -
-3
39
93
3.
.5
5 -
-3
39
94
4.
.4
4 2
21
13
3.
.6
6
Bromo
Bromo
CH
CH3
3C
CO
OO
OH
H(
(/
/)
) -
-4
48
87
7.
.0
0 -
-3
39
92
2.
.4
4 1
15
59
9.
.8
8
Br(g)
Br(g) 111.8
111.8 8
82
2.
.3
38
8 1
17
74
4.
.9
9 Cesio
Cesio
Br~(ac)
Br~(ac) -
-1
12
20
0.
.9 -
9 - 102.8
102.8 80.71
80.71 C
Cs
s(g)
(g) 7
76
6.
.5
50
0 4
49
9.
.5
53
3 1
17
75
5.
.6
6
Br
Br2
2(g)
(g) 3
30
0.
.7
71
1 3
3.
.1
14
4 2
24
45
5.
.3
3 Cs(D
Cs(D 2
2.
.0
09
9 0
0.
.0
03
3 9
92
2.
.0
07
7
Br2(0
Br2(0 0
0 0
0 152.3
152.3 Cs(s)
Cs(s) 0
0 0
0 85.15
85.15
H
HB
Br
r(
(g
g)
) -
-3
36
6.
.2
23
3 -
-5
53
3.
.2
22
2 1
19
98
8.
.4
49
9 CsCl(s)
CsCl(s) -
-4 4
4 42
2.
.8 - 4
8 - 41
14
4.
.4
4 101.2
101.2
Caldo
Caldo Cloro
Cloro
Ca(g)
Ca(g) 1
17
79
9.
.3
3 1
14
45
5.
.5 1
5 15
54
4.
.8
8 c
ci
i(g)
(g) 1
12
21
1.
.7
7 1
10
05
5.
.7 1
7 16
65
5.
.2
2
Ca(s)
Ca(s) 0
0 0
0 41.4
41.4 cr(oc)
cr(oc) -
-1
16
67
7.
.2
2 -
-1
13
31
1.
.2
2 5
56
6.
.5
5
CaCC^s, calata)
CaCC^s, calata) -
-1
12
20
07
7.
.1
1 -
-1
11
12
28
8.
.7
76
6 9
92
2.
.8
88
8 c
ci
i2
2(g
(g)
) 0
0 0
0 222.96
222.96
CaCl
CaCl2
2(s)
(s) -
-7
79
95
5.
.8
8 -
-7
74
48
8.
.1
1 1
10
04
4.
.6
6 HC
HCl
l(fl
(fle)
e) -
-1
16
67
7.
.2
2 -
-1
13
31
1.
.2
2 5
56
6.
.5
5
CaF
CaF2
2(s)
(s) -
-1
12
21
19
9.
.6 -
6 -1
11
16
67
7.
.3
3 6
68
8.
.8
87
7 H
HC
Cl
l(
(g
g)
) -
-9
92
2.
.3
30 -
0 -9
95
5.
.2
27
7 1
18
86
6.
.6
69
9
CaO(s)
CaO(s) -
-6
63
35
5.
.5
5 -
-6
60
04
4.
.1
17
7 3
39
9.
.7
75
5 C
Co
ob
ba
al
lt
to
o
Ca(OH)
Ca(OH)2
2(s)
(s) -
-9
98
86
6.
.2
2 -
-8
89
98
8.
.5
5 8
83
3.
.4
4 Co(g)
Co(g) 4
43
39
9 3
39
93
3 1
17
79
9
CaSO
CaSO4
4(
(s
s)
) -
-1
14
43
34
4.
.0
0 -
-1
13
32
21
1.
.8
8 1
10
06
6.
.7
7 Co(s)
Co(s) 0
0 0
0 28.4
28.4
1112
1112
- 2. A P É N D IC E C Cantid ades term odinám icas de sustancias selecci onad as a 298.15 K (25 °C) 11 13
AH j AGJ S° AH
°f AGj S°
Sustancia (kj/mol) (kj/mol) (J/mol-K) Sustancia (kj/mol) (kj/mol) (J/mo
Cobre FeCl3(s) -400 -334 142.3
Cu(g) 338.4 298.6 166.3 FeO(s) -271.9 -255.2 60.75
Cu(s) 0 0 33.30 F e ^ s ) -822.16 -740.98 89.96
CuCl2(s) -205.9 -161.7 108.1 Fe30 4(s) -1117.1 -1014.2 146.4
CuO(s) -156.1 -128.3 42.59 FeS2(s) -171.5 -160.1 52.92
CU20(S) -170.7 -147.9 92.36 litio
Cromo Li(g) 159.3 126.6 138.8
Cr(g) 397.5 352.6 174.2 Li(s) 0 0 29.09
Cr(s) 0 0 23.6 L iV ) -278.5 -273.4 12.2
Cr20 3(s) -1139.7 -1058.1 81.2 Li+(g) 685.7 648.5 133.0
Escandio
LiCl(s) -408.3 -384.0 59.30
Se(g) 377.8 336.1 174.7 Magnesio
Sc(s) 0 0 34.6 Mg(g) 147.1 112.5 148.6
Estroncio
Mg(s) 0 0 32.51
SrO(s) -592.0 561.9 54.9
MgCl2(s) -641.6 -592.1 89.6
Sr(g) 164.4 110.0 164.6
MgO(s) -601.8 -569.6 26.8
Mg(OH)2(s) -924.7 -833.7 63.24
Flúor
F(g) 80.0 61.9 158.7
Manganeso
F”((zc) -332.6 -278.8 -13.8
Mn(g) 280.7 238.5 173.6
F2(g) 0 0 202.7 Mn(s) 0 0 32.0
HF(g) -268.61 -270.70 173.51 MnO(s) -385.2 -362.9 59.7
MnOjís) -519.6 -464.8 53.14
Fósforo M n04“(ac) -541.4 -447.2 191.2
P(g) 316.4 280.0 163.2
P2(g) 144.3 103.7 218.1
Mercurio
P4(g) 58.9 24.4 280 Hg(g) 60.83 31.76 174.89
P4(s, rojo) -17.46 -12.03 22.85 Hg(0 0 0 77.40
P4(s, blanco) 0 0 41.08 HgCl2(s) -230.1 -184.0 144.5
PCI3(g) -288.07 -269.6 311.7 Hg2Cl2(s) -264.9 -210.5 192.5
PCl3(¡) -319.6 -272.4 217 Níquel
PF5(g) -1594.4 -1520.7 300.8 Ni(g) 429.7 384.5 182.1
PH3(g) 5.4 13.4 210.2 Ni(s) 0 0 29.9
P40 6(s) -1640.1 — — NiCl2(s) -305.3 -259.0 97.65
P4O10(s) -2940.1 -2675.2 228.9 NiO(s) -239.7 -211.7 37.99
POCl3(g) -542.2 -502.5 325
Nitrógeno
N(g)
po c i 3(o -5 97.0 - 520.9 222
472.7 455.5 153.3
H3P 04(ac) -1288.3 -1142.6 158.2
N2(g) 0 0 191.50
Hidrógeno NH3(ac) -80.29 -26.50 111.3
H(g) 217.94 203.26 114.60 NH3(g) -46.19 -16.66 192.5
h V ) 0 0 0 NH4+(ac) -132.5 -79.31 113.4
H+(g) 1536.2 1517.0 108.9 N2H4(g) 95.40 159.4 238.5
H2(g) 0 0 130.58 NH4CN(s) 0.0 — —
Hierro NH4C1(s) -314.4 -203.0 94.6
Fe(g) 415.5 369.8 180.5 NH4N0 3(s) -365.6 -184.0 151
Fe(s) 0 0 27.15 NO(g) 90.37 86.71 210.62
Fe2+(oc) -87.86 -84.93 113.4 NOj(g) 33.84 51.84 240.45
Fe3+(oc) -47.69 -10.54 293.3 N20(g) 81.6 103.59 220.0
FeCl2(s) -341.8 -302.3 117.9 N20 4(g) 9.66 98.28 304.3
- 3. 11 14 A P É N D IC E C Cantid ades termo dinám icas de sustan cias seleccion adas a 298.15 K (25 °C)
AHJ AGJ S° a h ; a g ; S°
Sustancia (kj/mol) (kj/mol) (J/mol-K) Sustancia (kj/mol) (kj/mol) (J/mol-K)
Nitrógeno (cont.) Selenio
NOCI(g) 52.6 66.3 264 H2Se(g) 29.7 15.9 219.0
HNO3(ac) -206.6 -110.5 146
n
HNO3(g) -134.3 -73.94 266.4
Si(g) 368.2 323.9 167.8
Oxígeno Si(s) 0 0 18.7
O(g) 247.5 230.1 161.0 SiC(s) -73.22 -70.85 16.61
O2(g) 0 0 205.0 Sici4(0 -640.1 -572.8 239.3
o 3(g) 142.3 163.4 237.6 SiO^s, cuarzo) -910.9 -856.5 41.84
OH-(oc) -230.0 -157.3 -10.7
Sodio
H20(g) -241.82 -228.57 188.83 Na(g) 107.7 77.3 153.7
H20 ( í) -285.83 -237.13 69.91 Na(s) 0 0 51.45
H A ® -1 36 .1 0 -10 5.48 2 32.9 Na+(í?c) -240.1 -261.9 59.0
H202(g) -187.8 -120.4 109.6 Na+(g) 609.3 574.3 148.0
Plata NaBr(ac) -360.6 -364.7 141.00
Ag(s) 0 0 42.55 NaBr(s) -361.4 -349.3 86.82
Ag+(«c) 105.90 77.11 73.93 Na2C0 3(s) -1130.9 -1047.7 136.0
AgCl(s) -127.0 -109.70 96.11 NaCl(flc) -407.1 -393.0 115.5
-31.05 -11.20 121.3
NaCl(g) -181.4 -201.3 229.8
AgjOís)
NaCl(s) -410.9 -384.0 72.33
AgNOsís) -124.4 -33.41 140.9
NaHC03(s) -947.7 -851.8 102.1
Plomo NaN 03(flc) -446.2 -372.4 207
Pb(s) 0 0 68.85 NaN03(s) -467.9 -367.0 116.5
PbBr2(s) -277.4 -260.7 161 NaOH(flc) -469.6 -419.2 49.8
PbCOj(s) -699.1 -625.5 131.0 NaOH(s) -425.6 -379.5 64.46
Pb(N03)2(«c) -421.3 -246.9 303.3 Na2OH4(s) -1387.1 -1270.2 149.6
Pb(N03)2(s) -451.9 — — Titanio
PbO(s) -217.3 -187.9 68.70 Ti(g) 468 422 180.3
Potasio Ti(s) 0 0 30.76
K(g) 89.99 61.17 160.2 TiCk(g) -763.2 -726.8 354.9
K(s) 0 0 64.67 riCi4(/) -804.2 -728.1 221.9
KCl(s) -435.9 -408.3 82.7 Tl02(s) -944.7 -889.4 50.29
KClOsís) -391.2 -289.9 143.0 Vanadio
KClO^flC) -349.5 -284.9 265.7 V(g) 514.2 453.1 182.2
K2C03(s) -1150.18 -1064.58 155.44 V(s) 0 0 28.9
KNO^s) -4 92 .7 0 -39 3.13 132.9
Yodo
K20(S) -363.2 -322.1 94.14
% ) 106.60 70.16 180.66
KOjís) -284.5 -240.6 122.5
n « :) -55.19 -51.57 111.3
K202(s) -495.8 -429.8 113.0
h(g) 62.25 19.37 260.57
KOH(s) -424.7 -378.9 78.91
I2(s) 0 0 116.73
KOH(flc) -482.4 -440.5 91.6
HI(g) 25.94 1.30 206.3
Rubidio Zinc
Rb(g) 85.8 55.8 170.0 Zn(g) 130.7 95.2 160.9
Rb(s) 0 0 76.78 Zn(s) 0 0 41.63
RbCl(s) -430.5 -412.0 92 ZnCl2(s) -415.1 -369.4 111.5
RbC103(s) -392.4 -292.0 152 ZnO(s) -348.0 -318.2 43.9