The Periodic Table Presentation 1
•Transferir como PPT, PDF•
21 gostaram•8,552 visualizações
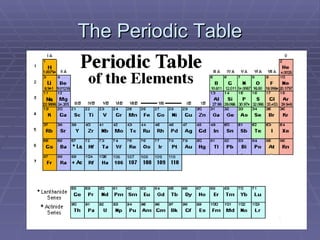
The document provides an overview of the development of the periodic table. It discusses early chemists like Lavoisier who compiled lists of known elements. In the 1860s, Newlands and Meyer began to notice patterns in properties of elements when arranged by atomic mass. Mendeleev later created the first periodic table by arranging elements in order of atomic mass with similar properties grouped together. This table had some issues resolved by Moseley in 1913, who arranged elements by atomic number, establishing the modern periodic table and periodic law.
Denunciar
Compartilhar
Denunciar
Compartilhar
Mais conteúdo relacionado
Mais procurados
Mais procurados (20)
Properties and Formation of Ionic Compounds Powerpoint

Properties and Formation of Ionic Compounds Powerpoint
Destaque
Destaque (20)
The periodic table presentation for 4050 [autosaved]![The periodic table presentation for 4050 [autosaved]](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![The periodic table presentation for 4050 [autosaved]](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
The periodic table presentation for 4050 [autosaved]
Moseley - His contribution To Physics and Chemistry

Moseley - His contribution To Physics and Chemistry
Semelhante a The Periodic Table Presentation 1
Semelhante a The Periodic Table Presentation 1 (20)
Periodic table of chemical elements impact on human welfare

Periodic table of chemical elements impact on human welfare
Chemistry Unit 2 Part 4 - Development and Organization of the Periodic Table

Chemistry Unit 2 Part 4 - Development and Organization of the Periodic Table
Mais de itamarita1984
Mais de itamarita1984 (20)
Último
Último (20)
Unraveling Multimodality with Large Language Models.pdf

Unraveling Multimodality with Large Language Models.pdf
Dev Dives: Streamline document processing with UiPath Studio Web

Dev Dives: Streamline document processing with UiPath Studio Web
What's New in Teams Calling, Meetings and Devices March 2024

What's New in Teams Calling, Meetings and Devices March 2024
Integration and Automation in Practice: CI/CD in Mule Integration and Automat...

Integration and Automation in Practice: CI/CD in Mule Integration and Automat...
WordPress Websites for Engineers: Elevate Your Brand

WordPress Websites for Engineers: Elevate Your Brand
Designing IA for AI - Information Architecture Conference 2024

Designing IA for AI - Information Architecture Conference 2024
Ensuring Technical Readiness For Copilot in Microsoft 365

Ensuring Technical Readiness For Copilot in Microsoft 365
Leverage Zilliz Serverless - Up to 50X Saving for Your Vector Storage Cost

Leverage Zilliz Serverless - Up to 50X Saving for Your Vector Storage Cost
"Subclassing and Composition – A Pythonic Tour of Trade-Offs", Hynek Schlawack

"Subclassing and Composition – A Pythonic Tour of Trade-Offs", Hynek Schlawack
Transcript: New from BookNet Canada for 2024: BNC CataList - Tech Forum 2024

Transcript: New from BookNet Canada for 2024: BNC CataList - Tech Forum 2024
"Federated learning: out of reach no matter how close",Oleksandr Lapshyn

"Federated learning: out of reach no matter how close",Oleksandr Lapshyn
Tampa BSides - Chef's Tour of Microsoft Security Adoption Framework (SAF)

Tampa BSides - Chef's Tour of Microsoft Security Adoption Framework (SAF)
Human Factors of XR: Using Human Factors to Design XR Systems

Human Factors of XR: Using Human Factors to Design XR Systems
Developer Data Modeling Mistakes: From Postgres to NoSQL

Developer Data Modeling Mistakes: From Postgres to NoSQL
Kotlin Multiplatform & Compose Multiplatform - Starter kit for pragmatics

Kotlin Multiplatform & Compose Multiplatform - Starter kit for pragmatics
The Periodic Table Presentation 1
- 1. The Periodic Table Part One
- 17. Problem-solving lab p 155