Postdoc Ph D Research Fei 2002
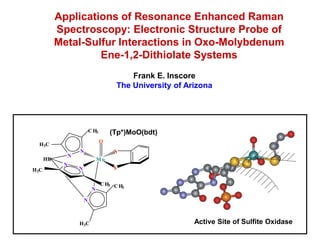
- 1. Applications of Resonance Enhanced Raman Spectroscopy: Electronic Structure Probe of Metal-Sulfur Interactions in Oxo-Molybdenum Ene-1,2-Dithiolate Systems Frank E. Inscore The University of Arizona C H3 (Tp*)MoO(bdt) O H3C N S N HB Mo N H3C N S C H3 C H 3 N N H3C Active Site of Sulfite Oxidase
- 2. The Raman Spectroscopic Technique General Considerations Research and Industrial Applications Structure/ Solid State/ Biological Chemistry Utility of Raman Spectroscopy Problematic: Inherent Weak Effect Problematic: Fluorescence Complications Problematic: Instrumental Limitations Development of New Techniques FT-RS/ SERS/ RERS
- 3. Resonance Raman Spectroscopy Characterizing Structure/ Monitoring Reactivity in Catalytic Systems Chemical and Petroleum/ Energy Production Industries • Catalyst Structure and Reactivity: Surface and In Situ Studies Heterogeneous processes: Supported metal oxides (MoO/ WO) used as catalyst. Hydrodesulfurization catalyst: Removal of sulfur from petroleum feedstocks. Biological Systems • Structure/ Function In Situ Studies Protonation in Biomolecules: S-H/ S-S conversion. Mechanistic insight into Carcinogenesis: Blue/green particle in tumors; Cu-S bonding. Structural Insight in Metalloproteins.
- 4. Overview of Presentation • Raman Applications • What we are doing? • Background • Electronic Structure • Why we are studying? • Raman Instrumentation • Resonance Raman Studies • How we will probe? • Implications for Mo Enzymes C H3 O H3C N S N HB Mo N H3C N S C H3 C H 3 N N H3C
- 5. The Importance of Metal 1,2-Dithiolene Complexes General Considerations Why the Interest in Transition Metal-Sulfur Complexes? Industrial Applications/ Commercial Uses: Vulcanization Accelerators for Rubber Wear Additive Inhibitors in Lubricants Catalytic Inhibitors /Oxidation Catalyst Mode-Locking Additives in Nd Lasers Potential Biological Activity: Correlations with Biological Systems containing Metal to Sulfur Bonds. What Is an Ene-1,2-Dithiolate Ligand ? Four Prototypical Ene-1,2-Dithiolate Systems: -S H -S -S N -S S -S M S = -S -S -S N -S S -S H S Relevance to structure, bonding and function of Metalloenzyme active site centers
- 6. Pyranopterin Molybdenum and Tungsten Enzymes Background and Significance 3 Mo Families based on structure and reactivity 2 W Families similar to DMSO reductase family O S O S er -O S O O Mo S S S Mo Mo S S - C ys S O H2 S S Sulfite Oxidase Xanthine Oxidase DMSO Reductase # X-ray crystallography reveals a common structural unit: Pyranopterin cofactor O S- H N S- HN H2N N N O OPO 2- H 3
- 7. The Resonance Raman Spectroscopic Probe Structure/ Bonding in the Active Site of DMSO Reductase Single Metal Redox Center RO O (VI) • XAS [Mo(VI,V,IV)] S S M • MCD/ EPR [Mo(V)] S S (Mo-S) • Electronic Absorption (Mo=O) x • Resonance Raman Observe enhanced isotopic sensitive Mo=O and Mo-S vibrations. Parallel model studies on both relevant and simpler systems needed.
- 8. Outstanding Issues in Pyranopterin Mo Enzyme Catalysis Primary Issue What is Structural and Functional Role of the Pyranopterin Ene-1,2- Dithiolate Unit During Course of Catalysis? Research Goal Derive fundamental understanding at molecular level, into how the unique geometric and electronic structure of these enzyme active sites contribute to their reactivity. Research Objectives Utilize available physical characterization methods to determine the geometric and electronic structure of small synthetic active site analogs. Derive key factors that define geometric/electronic structure relationships and correlate to the unique enzymatic spectroscopic features and their electronic contributions to structure-bonding/ function.
- 9. Chemical Evolution of Mo and W Dithiolene Systems The Reductionist Approach CH3 H3C O O -1, -2 O -1 N S RO HB N S S S S N N M M M H3C S S S S S CH3 -1 N CH3 OR N S S -1,0 M H3C S S S S CH3 M S S H3C S -2 N N Mo S OO HB S S H3C N N S M S S CH 0, +1 N 3CH3 N S M S -2 H3C OO CH3 S SR O M H3C 0, -1 S Y N N S HB N Mo O H3C N N S SS 0, -1, -2 -1 S SR S S M CH3 M S Y N CH3 S S N H3C
- 10. Minimal Structural Models/ Effective Spectroscopic Models Simple model; Mo coordinated by Ene-1,2-Dithiolate and terminal Oxo. Isolated Oxo-Mo-Dithiolate Center; Controlled six coordinate environment. Possess Mo(V) paramagnetic centers; Amenable to EPR/ MCD probes. C H3 E H3C N N S (S-S) HB M N S H3 C N Cl -S -S C H3 - S -S N CH C H 3 N 3 -S -S -S -S N N (Tp*)ME(S-S) Cl H3C Probe fundamental properties of Oxo-Mo mono-ene1,2-dithiolate complexes: Metal (M = Mo, W), axial (E = O, S, NO) and dithiolate (S-S) coordination effects.
- 12. MCD and Electronic Absorption Spectroscopy Low Temperature Solid-State (PDMSO Mull) Studies 0.8 1000 Absorption (5K) MCD (5K /7T) 0.7 0.6 500 MCD Intensity (mdeg) 0.5 Absorbance 0.4 0 0.3 0.2 1 2 -500 (3, 4) 5 6 7 0.1 0 -1000 8000 12000 16000 20000 24000 28000 32000 Energy (wavenumbers) Complimentary selection rules: resolve electronic transitions in spectra
- 13. Band Assignments from Combined Spectroscopic Approach y a'z2 Solution Electronic Absorption (DCE) y a''x2-y2 4 6400 y x'z y a'y'z a'' 7 3 6 7 5600 4800 y a'xy 1 2 5 Epsilon (M cm ) -1 4000 6 y a''op y a'op -1 3200 2400 y a'ip 5 1600 y a''ip 800 1 2 (3, 4) a'xy + a'ip a'xy + a'op a'yz + a'op O 0 M S 8000 12000 16000 20000 24000 28000 32000 = 90 0 Energy (wavenumbers) O M S > 90 0
- 14. Resonance Raman Scattering Enhancement of the Raman signal Sensitive and selective probe of structure/ bonding Vibrational frequencies: sensitive to inner coordination environment. Intensitiy: selective enhancement associated with absorbing metal center. Resonance FC - A Term Normal Raman Rayleigh Raman O HT - B Term E’ E1 o o M S O S IR ' M S E0 S Selectivity based on resonant electronic transition and excited state distortion. Intensity depends on energy and intensity of electronic absorption band. Enhancement result of coupling with electronic excited state.
- 15. Raman Experimental Instrumentation and Techniques Design and Methodology Goal: Obtain Low-frequency vibrational information regarding M-S bonding. Computer System Controller Interface Titanium Sapphire Laser Argon Ion Laser CCD Sample Krypton Ion Laser Illumination/ Collection Pre Monochromator Optics SPEX 1877 SPEX 1405 Triplemate
- 16. Collection Geometry Computer System Controller Interface Argon Ion Laser 90 degree geometry CCD Sample Illumination/ Collection Optics SPEX 1877 135 degree back scattering geometry Triplemate
- 17. The Resonance Raman Experiment
- 18. Laser Enhanced Raman Spectroscopy
- 20. Sample Illumination and Collection Optics
- 21. Sample Handling, Detection and Dispersal System Samples Problematic: Photo Decomposition/ Thermal Degradation?
- 22. Vibrational Raman Spectroscopy C H3 (Tp*)MoO(bdt) in NaCl/ Na2SO4 O 8500 H 3C 140K 528.7 nm ~40 mW N S 8000 N HB Mo 7500 3 N H3C N S 7000 1 C H3 C H 6500 6 Raman Intensity (cps) N 3 6000 N 5500 300 400 500 600 700 800 900 1000 1100 (Tp*)MoO(bdt) in Benzene H 3C 2000 293K 514.5 nm ~75 mW 1900 3 vibrational bands observed 1800 1700 1600 6 = 362 cm-1 1 = 393 cm-1 3 = 932 cm-1 1500 3 6 1 1400 1300 300 400 500 600 700 800 900 1000 1100 Raman-shift (wavenumbers) Identify normal modes coupled to electronic transitions
- 23. Solution Raman Depolarization Studies 2000 Depolarization Ratio (Tp*)MoO(bdt) in Benzene 1900 293K 496.5 nm ~75 mW 1800 = I/ I 1700 Parallel polarization 0 3/4 1600 I ¾ 1500 3 1400 6 1 Raman Intensity (cps) 1300 300 400 500 600 700 800 900 1000 1100 Totally symmetric (polarized) Non-totally symmetric (depolarized) 2000 1900 1800 Perpendicular polarization Ratio indicates 3 modes are totally symmetric 1700 6(A = 362 cm-1 ') 1600 I 1500 1(A = 393 cm-1 ') 1400 6 1 3 3(A = 932 cm-1 ') 1300 300 400 500 600 700 800 900 1000 1100 Raman-shift (wavenumbers)
- 24. Vibrational Analysis (Tp*)MoO(bdt) in Benzene 2000 CH3 O H3C O z z S NN S 1900 Mo N N Mo S HB S Raman Intensity (cps) H3C 1800 O O CH3 CH N 3 1700 N M y (zy) M y B N N S S 1600 S x S 362 cm-1 H C 3 932 cm-1 x N -1 1500 ( A' ) 393 cm ( A' ) 6 3 ( A' ) 1 1400 O O O 1300 300 400 500 600 700 800 900 1000 1100 M M M -1 S S S S S S Raman-shift (cm ) 1 ( A' ) 2 ( A'' ) 3 ( A' ) Key Points: 3 bands observed – polarized (A’ symmetry) O O O M M M Intensity enhancement patterns consistent S S S S S S With M-S/ M=O vibrational assignments 4 ( A' ) 5 ( A'' ) ( A' ) 6 Resonance Raman spectroscopy probes: Differences in bonding between ground and excited states via distortions along specific normal modes.
- 25. Solid-State Excitation Profiles Key Points: Observe large differential enhancement of Mo=O Transitions probed are orthogonal (in-plane vs out-of-plane) O (Tp*)MoO(bdt): 8K PDMSO mull EA; 100K RR NaCl/ Na2SO4 M S S 3( A' ) Sop Mo dxz,yz O Sip Mo dxy S M S 1( A' ) O M S S 6( A' ) Conclusions: Sip Mo dxy CT probes covalent contributions to ground-state S Mo CT probes electronic contributions to redox potentials
- 26. Implications for Catalytic Reactivity in Enzymes Lowest energy (intense) CT must be Sip Mo dxy This CT transition probes covalency contributions to ET pathway. M=O aligns redox orbital for facile ET via unique 3-center 2-electron bond. O SO32- SO42- O O OH 2 S S yxya' Mo (VI) Mo (IV) S S cys S S cys xya' H+, e- H2O S-Moxy 3-center H+, e- pseudo- antibonding O OH S-Moxy 3-center S pseudo- bonding Mo (V) a' ip S S cys Criteria for efficient ET yipa' Reason Nature has chosen ene-1,2-dithiolate and M=O groups Good M-L overlap/ Minimize ROE
- 27. Conclusions Resonance Raman Important Probe of Ground and Excited State Structure State of the Art Equipment Necessary for probing M-S Bonding. Contributions of M-L Bonding to Electronic Structure Elucidated by RR Especially when Combined with other Spectroscopic Techniques. RR Spectroscopy Important Tool for Characterizing Enzyme Active Sites when Interpreted within Context of Well-Defined Small Molecular Models. Protocols Developed can be Applied to more Complicated Systems.
- 28. Acknowledgements and Funding * HeI Prof. John H. Enemark HOMO Pseudo anti-bonding Mo dxy h = 579 nm Enemark Research Group HOMO -1&-2 HeII Sip University of Arizona Pseudo bonding * HOMO-3 &-4 * * 10.5 10 9.5 9 8.5 8 7.5 7 6.5 HOMO-5 Ionization Energy (eV) Prof. Martin L. Kirk National Institutes of Health National Science Foundation Kirk Research Group University of New Mexico Petroleum Research Fund Sandia National Laboratories C16 O C14 C15 S2 C4 C2 C13 MO C17 C3 C24 C26 S1 C5 N21 C1 C6 B N31 C23 N32 C36 (Tp*)M oO(qdt) in Benze ne at 514.5nm C27 C33 C37 C34 4000 (Tp*)MoO(bdt) 3800 Raman Intensity (cps) 3600 3400 (Tp*)WO(bdt) 3200 3000 10 9.5 9 8.5 8 7.5 7 6.5 6 400 600 800 1000 1200 1400 Ionization Energy (eV) -1 Ram an S h ift (cm)