Applications of-plant-tissue-culture
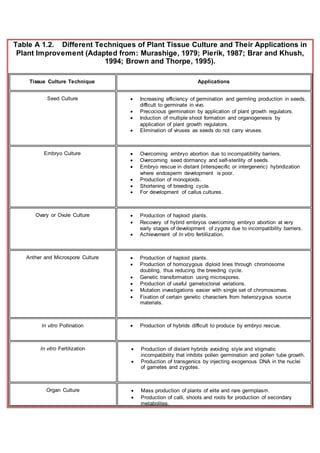
- 1. Table A 1.2. Different Techniques of Plant Tissue Culture and Their Applications in Plant Improvement (Adapted from: Murashige, 1979; Pierik, 1987; Brar and Khush, 1994; Brown and Thorpe, 1995). Tissue Culture Technique Applications Seed Culture Increasing efficiency of germination and germling production in seeds, difficult to germinate in vivo. Precocious germination by application of plant growth regulators. Induction of multiple shoot formation and organogenesis by application of plant growth regulators. Elimination of viruses as seeds do not carry viruses. Embryo Culture Overcoming embryo abortion due to incompatibility barriers. Overcoming seed dormancy and self-sterility of seeds. Embryo rescue in distant (interspecific or intergeneric) hybridization where endosperm development is poor. Production of monoploids. Shortening of breeding cycle. For development of callus cultures. Ovary or Ovule Culture Production of haploid plants. Recovery of hybrid embryos overcoming embryo abortion at very early stages of development of zygote due to incompatibility barriers. Achievement of In vitro fertillization. Anther and Microspore Culture Production of haploid plants. Production of homozygous diploid lines through chromosome doubling, thus reducing the breeding cycle. Genetic transformation using microspores. Production of useful gametoclonal variations. Mutation investigations easier with single set of chromosomes. Fixation of certain genetic characters from heterozygous source materials. In vitro Pollination Production of hybrids difficult to produce by embryo rescue. In vitro Fertilization Production of distant hybrids avoiding style and stigmatic incompatibility that inhibits pollen germination and pollen tube growth. Production of transgenics by injecting exogenous DNA in the nuclei of gametes and zygotes. Organ Culture Mass production of plants of elite and rare germplasm. Production of calli, shoots and roots for production of secondary metabolites.
- 2. Development of germplasm banks for rare and endangered plants. Shoot Apical Meristem Culture Production of virus free germplasm. Mass production of desirable genotypes. Facilitation of international exchange. Cryopreservation or In vitro conservation of germplasm. Phytosanitary transport. Somatic Embryogenesis Mass multiplication of elite germplasm. Production of artificial seeds. As source material for embryogenic protoplasts. For genetic transformation. Production of primary metabolites specific to seeds such as lipids in oil seeds. Amenable to mechanization and for bioreactors. Organognesis and Enhanced Axillary Budding Mass multiplication of elite germplasm. As source material for protoplast work, genetic transformation and mirografting. Conservation of endangered genotypes either at normal or at sub- zero temperatures. Callus Cultures Production of plantlets through somatic embryogenesis or organogenesis. For obtaining virus-free plants. For generation of useful somaclonal and gametoclonal variants. As a source of protoplasts and suspension cultures. Production of useful secondary metabolites. For biotransformation studies. Selection of cell lines with valuable properties such as resistance to disease, herbicides, overproduction of secondary metabolites etc. For mutagenetic studies. In vitro Production of Secondary Metabolites Production of useful compounds such as drugs, aromatic substances, pigments, flavors etc. without destruction of mother plants. Production of novel metabolites normally not produced by the parent plant. Biotransformation and elicitor studies. Cell Culture and In vitro Selection at Production of somatic embryos, morphognetic nodules and entire
- 3. Cellular Level plantlets. Over-production of secondary metabolites. Over-production of primary metabolites. Induction and selection of useful mutants or somaclones at cell level for disease resistance, stress tolerance and improved nutritional quality in less time and space. Somaclonal Variations (Genetic or Epigenetic) Isolation of useful variants in well-adapted, high yielding genotypes lacking in a few desirable traits. Isolation of useful variants overproducing primary or secondary metabolites. Isolation of useful variants with better disease resistance, stress tolerance capacities. Creation of additional genetic variation without hybridization in useful cultivars. In vitro Mutagenesis Induction of polyploidy for consequent increase in biomass or yield. Introduction of genetic variability and rapid selection as well as multiplication of useful mutants. As a tool for developmental genetics and for elucidation of biochemical processes. Protoplast Isolation, Culture and Fusion Combining distant genomes to produce somatic hybrids,asymmetric hybrids and cybrids. Production of organelle recombinants. Transfer of CMS (cytoplasmic male sterility) in elite lines. Source material for genetic transformation. Creation of genetic variants. Genetic Transformation Introduction of foreign DNA to generate novel genetic combinations. Transfer of desirable genes for disease and pest resistance from related or unrelated plant species into high yielding susceptible cultivars. Study of structure and function of genes. Induction of hairy roots or shooty terratomas for over-production of secondary metabolites, naturally present in mother plant. Production of novel secondary metabolites absent in parent plant. In vitro Flowering Reduction in long life cycle in perennials such as Bamboo. Continuous supply of flowers, fruits and seeds irrespective of season. Micrografting Overcoming graft incompatibility. Rapid mass propagation of elite scions grafted on rootstocks having desirable traits like resistance to soil-borne pathogens and diseases. Multiplication and survival of difficult to root species as well as of transformants.
- 4. Development of virus free plants. Cryopreservation or Storage at Low Temperature Long term preservation of useful germplasm (cell lines, meristems, plant organs, morphogenetic callus cultures). Conservation of natural genetic variability. Culture of protoplasts, cells, tissues and organs As a tool in Phytopathological Research. Virus preparation and replication. Culture of obligate parasites. Host-parasite interactions. Culture of nematodes (Excised root cultures). Testing of phytoalexins and phytotoxins. Nodulation studies. As a tool in Plant Physiological Research. Cell cycle studies. Metabolic studies. Nutritional studies. Morphogenetical and developmental studies. Culture of hairy roots For understanding and manipulating root-specific metabolism. For co-culture with VAM fungi to increase secondary metabolite production. For co-culture with insects to study pathogenesis. For study and commercial exploitation of bioactive root exudates. For co-culture with shooty teratomas to exploit both root and shoot based metabolism for biotransformations and also for metabolite production specific with both these locations. For development of "green hairy roots" which are photoautotrophic and hence display a different spectrum of secondary metabolites.
