Nicolaou
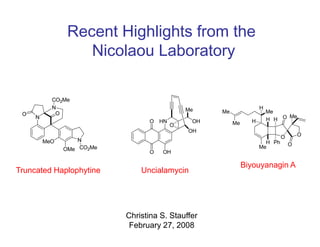
- 1. Recent Highlights from the Nicolaou Laboratory Me H Me Me Me O HN OH H H H O O Me OH O O H Ph O Me O OH Biyouyanagin A Truncated Haplophytine Uncialamycin Christina S. Stauffer Ch i ti S St ff February 27, 2008
- 2. Haplophytine Me N O O N N HO O MeO N O H OMe Me Haplophytine p p y CO2Me N N O O N 9 9' O 15 MeO N O H N OMe Me MeO OMe CO2Me Aspidophytine Truncated haplophytine Syntheses: Corey, 1999 Fukuyama, 2003 Padwa, 2006 Marino, 2006 • Potent insecticidal alkaloid alkaloid. • Isolated in 1952 by Snyder and co-workers from Mexican plant Haplophyton cimicidum. • Consists of a central indole moiety with two tetracyclic heterocycles attached. attached • Four reported syntheses of aspidophytine, while only one report of studies towards the left-hand domain (Corey, 2006).
- 3. Retrosynthesis of the “Left Domain” of Haplophytin CO2Me N O O N 9' 15 MeO M O N OMe CO2Me Truncated haplophytine N N CO2Me H HO N OH CO2Me CO2Me Nicolaou, K. C.; Majumder, U.; Roche, S. P.; Chen, D. Y.-K. Angew. Chem. Int. Ed. 2007, 46, 4715-4718.
- 4. Synthesis of indole fragment NH NCCO2Me, CH2Cl2, 0 °C N N H N CO2Me 51% H CO2Me CO2Me
- 5. Synthesis of the indoline fragment CHO 1. Ac2O, Et3N, cat. DMAP CHO 2. fuming HNO3 HO HO NO2 3. aq. KOH, reflux OMe OMe 69% 1. K2CO3, BnBr, DMF 2. CH3NO2, NaOH, , NO2 1. Fe, AcOH, tol, 110 °C MeOH/H2O, 0 °C 2. KHMDS, NCCO2Me, 3. Ac2O, NaOAc, 140 °C BnO NO2 THF, -78 °C 88% OMe 96% 1. Pd/C, H2, MeOH , HO N BnO N 2. BBr3, CH2Cl2, -78 °C to 0 °C OH CO2Me OMe CO2Me 86%
- 6. Coupling of indole and indoline fragments N CO2Me PIFA = PhI(CF3CO2)2 OH O CH2Cl2, CH3CN, -40 °C N N CO2Me HO N O H 25% OH CO2Me N CO2Me N CO2M Me H CO2Me N CO2Me N CO2Me OMe OM OMe Cs2CO3, MeI DMF OMe NaH, THF OMe 60% N 80% N N CO2M Me N CO2Me O CO2Me
- 7. Completion of the construction of the “left domain” of haplophytin
- 8. Uncialamycin Me O HN OH O OH O OH Cladonia uncialis • A recently discovered enediyne antibiotic isolated in 2005 by Anderson from the surface of the lichen, Cladonia uncialis. • The scarcity of uncialamycin (300 μg) limited structural elucidation (C26 hydroxyl group was unassigned). • Potent biological activity demonstrated against S. aureus, E. coli, and B. cepacia.
- 9. Nicolaou’s retrosynthesis of uncialamycin Me O HN OH O OH O OH Me CN N O O OTES H TMS O ODMB Nicolaou, K. C.; Zhang, H.; Chen, J. S.; Crawford, J. J.; Pasunoori, L. Angew. Chem. Int. Ed. 2007, 46, 4704-4707.
- 10. Synthesis of the substituted quinoline system O Me HN O CO2H OMe O Me 1. 48% aq. HBr, N Me HCO2H, Et3N, N nBu4NBr, 110 °C O CH2Cl2, 0 °C CO2DMB 2. DMBBr, K2CO3, O nBu4NI, DMF Me Me ODMB ODMB 55% Ts Ph N Ru N Me Ph H2 Cl 95%, 98% ee Me OMe N 1. DIBAL-H, CH2Cl2 O O DMB = 2. TESCl, im, DMF OTES Ph OMe 86% OR ODMB
- 11. Introduction of the enediyne functionality TMS Me Me TMS 1. AcOH, MeCN, Me N AllocN H2O, 91% O H TMS O 2. NaBH4, MeOH AllocN OH O OTES EtMgBr, AllocCl OTES 3. mCPBA, CH2Cl2 92% 80% (2 steps) OAc ODMB ODMB 4. AcCl, collidine, 82% ODMB TMS H Me 1. TESCl, Im, Me 1. Dess-Martin AllocN OH DMF O AllocN OTES O 2. 2 NaBH4, MeOH 2. 2 K2CO3, THF OAc MeOH 90% OH 78% ODMB ODMB
- 12. Intramolecular acetylide addition to form the enediyne ring system H H Me 1. Dess-Martin, 87% Me CeCl3, KHMDS - 78 °C to - 40 °C AllocN OTES 2. DDQ, 2 DDQ CH2Cl2/H2O AllocN OTES O O 61% 87% (30% C17 epimer) OH O ODMB OH Me Me 1. PhI(OAc) MeOH 1 PhI(OA )2, M OH AllocN OTES 80% N OTES CN O 17 O OH OH 1. 2. [Pd(PPh3)2Cl2] O nBu3SnH, H2O, CH2Cl2, 74% O OH O LiHMDS, 63% 2. 3HF•Et3N, THF 92% Me O HN OH O OH O OH Uncialamycin
- 13. Synthesis of Biyouyanagin A (H. chinense L. var. salicifolium) • Isolated from the leaves of a Hypericum species in 2005. • Traditionally used as folk remedy for female disorders. • Shows significant activity against HIV and lipopolysaccharide-induced cytokine production. • Stereochemistry at C24, C17, C18 needs to be elucidated.
- 14. Structural assignment of biyouyanagin A H H Me Me Me Me Me Me H H H O H H H O Me Me O O O O H Ph O H Ph O Me Me Originally proposed possible structures H H Me Me Me Me Me H H H O Me H 24 H H O Me Me 17 18 O O O O H Ph H Ph O Me O Me Nicolaou, K. C.; Sarlah, D.; Shaw, D. M. Angew. Chem. Int. Ed. 2007, 46, 4708-4711.
- 15. Biyouyanagin A retrosynthesis H H Me Me Me Me Me O Me H H H O Me H Me [2 + 2] O O O O Ph H Ph O Me O Me
- 16. Synthesis of the terpene-derived diene Cl Tf N N Tf Comins reagent
- 17. Formation of the spirolactone
- 18. [2+2] Photocycloaddition H H Me Me Me Me O Me Me H h , 2'-acetonaphthone H H H O Me Me CH2Cl2, rt, 5h O O O Ph 46% O O H Ph O Me Me H Me Me H Me OO Me O O Me (exo)