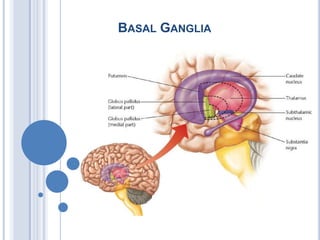
Basal Ganglia Clinical Anatomy Physiology
- 2. SUBDIVISION OF BG A. Neostriatum or Striatum Putamen Caudate nucleus B. Pallio striatum or Pallidum Globus pallidus C. Lentiform nucleus Putamen Globus Pallidus D. Archistiatum Amygdela E. Substantia nigra F. Subthalamic nucleus
- 3. NERUONS AND CIRCUITS OF BG
- 5. MOTOR COMPONENT OF BASAL GANGLIA
- 7. BG EFFERENTS
- 8. CONNECTIONS OF THE NEOSTRIATUM WITH THE SUBSTANTIA NIGRA
- 9. CORTICAL LOOP Cortex: 4,6,temporo, parietal and occipital glutamate Striatum spiky GABA Pallidum medial GABA Thalamus VL, VA
- 10. BG: SUBCORTICAL LOOPS In the case of all sub-cortical loops the position of the thalamic relay is on the input side of the loop Red – Excitatory Blue - Inhibitory
- 11. INTERNAL CONNECTIONS OF THE BASAL GANGLIA: DIRECT PATHWAY
- 13. PROJECTIONS FROM THE BASAL GANGLIA TO OTHER BRAIN REGIONS
- 14. FUNCTION OF BG Voluntary movement Initiation of movement Control of ramp movement Change from one pattern to other Programming and correcting movement while in progress (thalamocortical circuts) Postural control Righting reflex Automatic associated movement (walking) Control of muscle tone Reticulospinal Vestibulospinal
- 15. ABLATION STUDY Unilateral lesion minimal effect Denny Brown – Bilateral lesion → Akinesia + Flexion dystonia Brook – Cooling of GP → Contralateral cocontraction of antagoniastic muscle → Flexion, alternate and amplitude of movement Bilateral striatal ablation → Overactive, does not respond to visual cue – walk to wall Bilateral pallidal ablation → Hypoactive akinetic Human sterotactic Gpi lesion → tremor >rigidity Subthalamic Nucleus ablation → Hemibalismus Bilateral CN ablation → immobile animal VL thalamic cooling → Ia discharge to stretch reflex → rigidity by y tone
- 16. STIMULATION STUDIES CN stimulation → head and body turn to opposite site, circling movement, or mild hypertonia, late tremor, changes tonic to clonic phase of epilepsy Neostriatal stimulation → arrest of motion in progress
- 17. MICROELECTRODE RECORDING Activity seen during initiation of internally generated movement but not to stimulus triggered movement Activity seen during co-contraction of agonist (stimulus triggered thus control amplitude and velocity of movement Preparation of motor act or programming as MC and SMA React Spontaneous movement of individual body part Alternating movement Visually and kinesthetically triggered movement Postural adjustment to body tilt Rapid ballistic movement Slow ramp movement Isometric muscle contraction SNc – tonic discharge -> postural control SNr - phasic discharge change with limb movement
- 18. Cortex DISCHARGE OF MOTOR CIRCUIT No spontaneous discharge, only during limb movement increased phasic activity High spontaneous discharge, inhibit tonically thalamus Phasic reduction of activity during movement due to disinhibition by striatum Phasic activity during movement Striatum GPi SNr Thalamus 1 2 3 4 5
- 19. BG AND EYE MOVEMENT
- 21. POSSIBLE ROLE OF INTRINSIC CIRCUITS
- 23. DISORDER OF BASAL GANGLIA Disorder of voluntary movement Bradykinesia Rapid alternating movement Intension spasm Postural abnormalities Gait disorder Tone changes Involuntary movement Phonation, articulation
- 24. AKINESIA AND BRADYKINESIA Akinesia is disorder characterized by poverty and slowness of initiation and execution of willed and associated movement and difficulty in changing one motor patter to another, in absence of paralysis. Disinclination to use a part Bradykinesia: Slow execution of movement due to failure of development of initial burst (low amplitude agonist burst) during ballistic movement. Selection of muscles and agonist : antagonist relationship is retained. The final position is often achieved by a repetitive series of small agonist burst → sequence of incremental steps
- 25. MECHANISM OF BRADYKINESIA Putamen → Globus pallidus internus encodes amplitude information during movement Increased reaction time for initiation of movement and correcting or stopping movement Velocity of movement reduced due to defective programming of movement both ramp and ballistic and disorderly recruitment of motor unit by pyramidal tract (thalamo-cortical circuit ↓) Not due to rigidity Defect in message from basal galngia to SMA →MC
- 26. MECHANISM OF BRADYKINESIA CONT… Difficulty in generating smooth continuous movement of all kind due to failure of generator function Changes in cerebral cortex secondary to basal ganglia Impairment of nigro-striatal dopaminergic system, improved by L-dopa Caudate n. stimulation → inactivation syndrome Globus pallidus cooling → akinesia Meso-limbic dopaminergic pathway abnormality of ventral tegmental tract → nucleus accumbence may be responsible Pre-movement potential reduced proportional to akinesia
- 27. MECHANISM OF BRADYKINESIA CONT… Tegmental dopamine lesion → continuous synchronized burst of Gpi. Reduced by Dopamine or Apomorphine Also disturbance of phasic changes in discharge → un-modifiable inhibition of thalamic neuron due to sustained unchanging discharge of Gpi. Normal motor plan but inaccurate in-exact specification and initiation of agonist activity and running smooth sequence of motor programes
- 28. HASTENING PHENOMENON Proportional to akinesia but not to tremor and rigidity Defective checking Not controlled by L-dopa, or thalamotomy
- 29. FREEZING PHENOMENON Not related to akinesia Appears 5-6 years after L-dopa therapy Due to ↓ NA in frontal cortices –striatal complex Dopamine beta hydroxylase in CSF reduced L-threo-DOPS (L-threo -3-4 dihydroxyphenyl serine) precursor of NA improve Can walk up and down (visual and auditory modality) Related to frontal apraxia
- 30. POSTURAL ABNORMALITY Involuntary flexion of trunk limb and head Inability to correct while turning limb and head Inability to correct while turning /falling Anticipatory postural reflex that precede shift of center of gravity are abnormal due to GP lesion → failure of compensatory righting reflex Whether GP or more diffuse abnormality ?
- 31. ALTERATION OF MUSCLE TONE Hypertonia Gamma loop over activity - ↑ long loop tonic stretch reflex activity release by basal ganglia brain stem and cord αγ co-contraction → rigidity Relived by Thalamotomy or Gpi lesion Hypotonia Degeneration of micro-neuron of thalamus with chorea
- 34. THE END
Notas do Editor
- Figure 20-3 Relationship of the cerebral cortex to the putamen and caudate nucleus. The diagram illustrates that the putamen receives fibers from motor regions of the cerebral cortex, while the caudate nucleus receives inputs from association regions (temporal and parietal lobes) as well as inputs from other regions of the frontal lobe, including the prefrontal cortex.The largest afferent source of the basal ganglia arises from the cerebral cortex. In fact, most regions of the cortex contribute projections to the basal ganglia. These include inputs from motor, sensory, association, and even limbic areas of the cortex. While the caudate nucleus and putamen serve as the primary target regions of afferent projections from the cortex, the source of cortical inputs to these regions of the basal ganglia differ. The principal inputs from the primary motor, secondary motor, and primary somatosensory regions of cortex are directed to the putamen. These inputs to the putamen are somatotopically organized, which means that different regions of the putamen receive sensory and motor inputs that are associated with different parts of the body. The caudate nucleus, on the other hand, receives inputs from cortical association regions, frontal eye fields, and limbic regions of cortex (Fig. 20-3). Thus, the putamen appears to be concerned primarily with motor functions, while the caudate nucleus, which appears to receive more varied and integrated cortical inputs, is likely involved with cognitive aspects of movement, eye movements, and emotional correlates of movement (relegated to the ventral aspect of the neostriatum).The neostriatum also receives an indirect source of cortical input. The source of this input is the cen-tromedian nucleus of the thalamus. This nucleus receives afferent fibers primarily from the motor cortex and projects its axons topographically to the putamen as thalamostriate fibers. An additional but highly important source of dopaminergic inputs to the neostriatum is the substantia nigra. The function of this afferent source to the neostriatum is considered later in this chapter.The modular organization of the neostriatum is as follows. The projections from the neocortex and thalamus are not distributed uniformly to the neostriatum. Instead, the projections end in compartments within different parts of the neostriatum. The smaller of these compartments is referred to as a patch or striosome and is surrounded by a larger compartment referred to as a matrix. These two compartments are distinguished from each other because they have different neurochemical properties, contain different receptors, and receive inputs from different cortical regions. The matrix is acetylcholinesterase rich. The striosomes, however, are acetylcholinesterase poor but contain peptides such as somatostatin, substance P, and enkephalin. The striosomes receive their inputs mainly from limbic regions of cortex, and neurons from this region preferentially project to the substantia nigra. In contrast, the neurons of the matrix receive inputs from sensory and motor regions of cortex, and many of them project to the globus pallidus. The overwhelming majority of projection neurons from both of these compartments of the neostriatum are γ-aminobutyric acid (GABA)-ergic. Their significance is considered in the following paragraph.The presumed role of the neostriatum in motor functions can be illustrated by the following example. During active movement of a joint of a finger, the cells within a certain striosomal or matrix compartment become active, while neurons embedded within other compartments become active only after there is passive movement of the same joint. This would suggest that the neurons embedded within given compartments function in a similar manner to those present in functional homogenous regions of the cerebral cortex with respect to movement (i.e., specific regions of motor cortex can be subdivided into distinct functional columns in which the neurons in one column respond to one feature of movement of a limb, whereas those neurons situated in an adjoining column respond to a different feature of movement of that limb;
- Figure 20-5 Key relationships of the substantia nigra. The pars reticulata of the substantia nigra receives an inhibitory (red line) GABAergic input from the neostriatum. In turn, there are two important outputs of the substantia nigra. The first is a dopaminergic (DA) projection to the neostriatum (which is excitatory when acting through D1 receptors and inhibitory when acting through D2 receptors). The second is an inhibitory GABAergic projection from the pars reticulata to the ventral anterior (VA) and ventrolateral (VL) thalamic nuclei as well as to the superior colliculus. +, excitation; -, inhibitionConnections of the Neostriatum with the Substantia NigraThe substantia nigra has two principal components: a region of tightly compacted cells, called the pars compacta, and a region just ventral and extending lateral to the pars compacta, called the pars reticulata (Fig. 20-2B). Fibers arising from the neostriatum project to the pars reticulata. Transmitters identified in this pathway are GABA and substance P. The pathway from the substantia nigra to the neostriatum arises from the pars compacta and uses dopamine as its neurotransmitter (Fig. 20-5). The pars reticulata also gives rise to efferent fibers (which are likely inhibitory) that project to the thalamus, superior colliculus, and locally to the pars compacta. In this manner, the pars reticulata and internal pallidal segment appear to be functionally analogous because both regions provide outputs to the cerebral cortex via the thalamus. Additional functions of these pathways are discussed later in this chapter.
- Figure 4: Cortical and subcorticalsensorimotor loops through the basal ganglia. A. For cortico-basal ganglia loops the position of the thalamic relay is on the return arm of the loop. Predominantly excitatory regions and connections are shown in red while inhibitory regions and connections are blue. Abbreviations: Thal, thalamus; SN/GP, substantia nigra/globus pallidus.Cortical-loopsManifest topographies associated with input projections, intrinsic connections and outputs of the basal ganglia provided a basis for the influential organisational principle suggested by Alexander and colleagues (1986). Connections between the cerebral cortex and basal ganglia can be viewed as a series of parallel projecting, largely segregated cortico-striato-nigro-thalamo-cortical loops or channels (Figure 3). Thus, an important component of the projections from different functional territories of cerebral cortex (e.g. limbic, associative, sensorimotor) project to exclusive functional territories in the basal ganglia input nuclei, which are then maintained in the internal circuitry. Output signals from functional territories represented in the output nuclei are returned, via appropriate thalamic relays, to the cortical regions providing the original input signals (Middleton and Strick 2000).
- Figure 4: B. In the case of all sub-cortical loops the position of the thalamic relay is on the input side of the loop. Predominantly excitatory regions and connections are shown in red while inhibitory regions and connections are blue. Abbreviations: Thal, thalamus; SN/GP, substantia nigra/globus pallidus.Sub-cortical loopsThe concept of potentially segregated parallel projecting loops through the basal ganglia has been extended to their connections with sensorimotor and motivational structures in the brainstem, including the superior colliculus, periaqueductal grey, pedunculopontine and parabrachial nuclei (McHaffie et al. 2005). An important difference is that, in the case of cortical loops, the thalamic relay is on the output side of the loop, whereas for the sub-cortical loops the thalamic relay is on the input side (Figure 4). Much work will be required to test whether projections from different brainstem structures, as they pass through the thalamic and basal ganglia relays, represent functionally segregated channels.
- Figure 20-4 The detailed anatomical and functional nature of the input, internal, and output circuits of the basal ganglia. Note the distinction between the direct and indirect pathways. (A) The direct pathway involves projections from the neostriatum to the medial (internal) pallidal segment, which in turn, projects to the thalamus and then to the cerebral cortex. Internal Connections of the Basal GangliaThe anatomical relationships between different components of the basal ganglia are extensive. The most salient of the connections include the following: (1) the projections from the neostriatum to the globus pallidus; (2) the reciprocal relationships between the neostriatum and substantia nigra; and (3) the reciprocal relationships between the globus pallidus and the subthalamic nucleus. In examining these relationships, the overall role of the basal ganglia in motor functions should be kept in mind. Namely, as signals are transmitted through the basal ganglia in response to cortical inputs, they ultimately result in a distinct response transmitted back to the motor areas of the cerebral cortex. Moreover, the circuits within the basal ganglia by which signals are transmitted back to the cerebral cortex may be direct or indirect. The differences between the direct and indirect routes are discussed in the following section.Connections of the Neostriatum with the Globus PallidusThere are two basic projection targets of the neostriatum: the globus pallidus and the substantia nigra. The neostriatum projects to two different regions of the globus pallidus: the medial (internal) pallidal segment and the lateral (external) pallidal segment (Fig. 20-4). GABA mediates the pathway from the neostriatum to the medial pallidal segment; likewise, the pathway from the neostriatum to the lateral pallidal segment also primarily uses GABA as a likely neurotransmitter.Each of these projections forms the initial links of two different circuits within the basal ganglia. Because the primary output of the basal ganglia is from the internal pallidal segment, the projection (neostriatum → globus pallidus (internal) → thalamus → neocortex) is called the direct pathway (Fig. 20-4). In contrast, the external segment of the globus pallidus shares reciprocal projections with the subthalamic nucleus. Thus, this circuit of the basal ganglia can be outlined as follows: neostriatum → globus pallidus (external) → subthalamic nucleus → globus pallidus (internal) → thalamus → neocortex). Because this circuit involves a loop through the subthalamic nucleus, it is called the indirect pathway (Fig. 20-4). The neurotransmitters involved in the reciprocal pathway between the globus pallidus and subthalamic nucleus and the overall functional significance of these pathways are discussed later in this chapter.
- (B) The indirect pathway involves projections from the neostriatum to the lateral pallidal segment (L), which in turn, projects to the subthalamic nucleus. The subthalamic nucleus then projects to the medial pallidal segment (M), and the remaining components of the circuit to the cerebral cortex are similar to that described for the direct pathways. Red arrows depict inhibitory pathways, and blue arrows indicate excitatory pathways. The green pathways represent dopaminergic projections to the neostriatum, which have opposing effects on D1 and D2 receptors (see Fig. 20-5), and the excitatory projection from the subthalamic nucleus (Sth) to the medial pallidal segment. When known, the neurotransmitter for each of the pathways is indicated. Abbreviations: DA, dopamine; GLU, glutamate; CM, centromedian nucleus; VA, ventral anterior nucleus; VL, ventrolateral nucleus; +, excitation; -, inhibition.
- Selecting and maintaining purposeful motor activity while suppressing unwanted or useless movement. Helping monitor and coordinate slow, sustained contractions related to posture and support. Inhibiting muscle tone throughout the body (proper muscle tone is normally maintained through a balance of excitatory and inihibtory inputs to the neurons that inervate skeletal muscle).
- The role of basal ganglia disinhibition in the generation of saccadic eye movements. (A) Medium spiny cells in the caudate nucleus respond with a transient burst of action potentials to an excitatory input from the cerebral cortex (1). The spiny cells inhibit the tonically active GABAergic cells in substantia nigra pars reticulata (2). As a result, the upper motor neurons in the deep layers of the superior colliculus are no longer tonically inhibited and can generate the bursts of action potentials that command a saccade (3,4). (B) The temporal relationship between inhibition in substantia nigra pars reticulata (purple) and disinhibition in the superior colliculus (yellow) preceding a saccade to a visual target. (After Hikosaka and Wurtz, 1989.)
- Figure 8: Potentially converging inputs to the dorsal striatum at the time of an unpredicted biologically salient visual event. A. Phasic sensory: Two separate short-latency representations of the visual event could converge on striatal circuitry: (i) retino-tecto-thalamo-striatal projections will provide a phasic sensory-related glutamatergic input (red arrows) ; and (ii) retino-tecto-nigro-striatal projections will provide a phasic dopaminergic input (orange arrows) . B. Contextual: Striatal neurones are sensitive to experimental context. Multidimensional contextual afferents are likely to originate from cerebral cortex, limbic structures (hippocampus and amygdala) and the thalamus (Blue arrows). C. Motor copy: Branched pathways from motor cortex and subcorticalsensorimotor structures (e.g. superior colliculus) reach the striatum directly (cortex) or indirectly via the thalamus (subcortical structures). Motor-related projections could provide the striatum with a running, multidimensional record (motor efference copy) of commands relating to ongoing goals/actions/movements (green arrows).Reinforcement learningThe basal ganglia have long been associated with the processes of reinforcement learning (Schultz 2006; see also Reward Signals). This should not be surprising since instrumental or operant conditioning (the class of learning most commonly linked to the basal ganglia) can be viewed as the biasing of future action selections by past action outcomes. One of the strongest lines of evidence supporting the involvement of the basal ganglia in reinforcement learning is the electrophysiological data obtained from behaving monkeys. Typically, unexpected biologically significant events including sudden novel stimuli, intense sensory stimuli, primary rewards, and arbitrary stimuli classically conditioned by association with primary rewards evoke a stereotypic sensory response from DA neurones in many species (Schultz 1998). This response comprises a characteristic short latency (70-100 ms), short duration (<200 ms) burst of activity. However, it is the capacity of phasic DA responses to change when experimental conditions are altered that has provoked most interest. The novelty response of DA neurones habituates rapidly when a sensory stimulus is repeated in the absence of behaviourally rewarding consequences. A phasic DA response will emerge following the presentation of a neutral sensory stimulus that predicts a primary reward. Under these conditions the DA responses to the predicted reward gradually diminish. When a predicted reward is omitted, a reliable depression in the spontaneous activity of the DA neurones occurs 70-100 ms after the time of expected reward delivery. It is largely on the basis of these data that the reward-prediction error hypothesis was originally formulated. More recently, additional supporting investigations have established that the phasic DA signal complies with the contiguity, contingency and prediction error tenets of contemporary learning theories (Schultz 2006). This body of evidence provides powerful support for the reward prediction error hypothesis which is now widely accepted by both biological and computational neuroscientists. Within this framework, the hypothesised errors in reward prediction signalled by phasic dopamine activity are presumed teaching signals for appetitive learning and ensure that actions maximising the future acquisition of reward are selected more often. However, recent evidence from studies that have identified sources of short-latency sensory input to midbrain dopaminergicneurones suggests that, in real world conditions where unexpected stimuli are both temporally and spatially unpredictable, the identity of unexpected events (and hence their reward value) will be determined after, rather than before the time of phasicdopaminergicsignalling (Redgrave and Gurney 2006). Although dopamine neurones have reliable responses to reward-related stimuli they also exhibit strong phasic responses to unexpected sensory events that have no obvious appetitive reinforcement consequences (Horvitz 2000). Despite reward-related stimuli coming in all sorts of shapes and sizes, the phasic dopamine signal is highly stereotyped (latency 100 ms, duration 100 ms) and largely independent of animal species, stimulus modality, and perceptual complexity of eliciting events (Schultz 1998). The 100 ms response latency of dopaminergicneurones is reliably shorter than the latency of the gaze-shift that brings the unexpected event onto the fovea for detailed analysis by cortical visual systems. Necessarily this means that dopamine responses are triggered as a consequence of limited pre-attentive, pre-saccadic sensory processing (Redgrave and Gurney 2006). Recent evidence indicates that the sensory inputs to dopaminergicneurones derive largely, if not exclusively as a consequence of early, subcortical sensory processing (Redgrave and Gurney 2006). In the case of vision, the midbrain superior colliculus is configured to indicate where an unexpected event is rather than what it is (Wurtz and Albano 1980). Perhaps it is no coincidence that, in almost all studies showing phasic dopamine signals can signal reward prediction errors (Schultz 2006), the economic values predicted by the conditioned stimuli are correlated with the spatial location of stimulus presentation. It therefore remains to be determined whether dopamine neurones can signal continuous values of reward prediction errors in real world conditions where unexpected events are both temporally and spatially unpredictable.
- Figure 20-8 Schematic diagram depicting the possible mechanisms for hypokinetic and hyperkinetic disorders. (A) Normal input-output relationships of the basal ganglia. Abbreviations: GLU, glutamate; GPL, lateral pallidal segment; GPM, medial pallidal segment; SN, substantia nigra; Sth, subthalamic nucleus; VA, ventral anterior; VL, ventrolateral.The information presented thus far has enabled us to identify the major circuitry of the basal ganglia. This includes the primary input pathways and the internal circuitry of the basal ganglia and their output pathways. In terms of the functional properties of this system, they may be best understood by following the flow-through relationships beginning with the inputs from the cerebral cortex.As noted earlier, there are both direct and indirect pathways from the striatum to the thalamus, and the functional effects of each are different. Concerning the direct pathway, cortical inputs have excitatory effects on the neostriatum, and the neurotransmitter is glutamate. Excitation of the neo-striatum by the cortex results in disinhibition of thalamic nuclei. This can be understood in terms of the following relationships. The projection from the neostriatum to the medial pallidal segment is inhibitory, and likewise, the projection from the medial pallidal segment to the thalamus is also inhibitory. GABA mediates both inhibitory pathways. In this manner, activation of the cortex causes the neurons in the medial pallidal segment to be inhibited by the GABAergic projection from the neostriatum. When the neurons of the medial pallidal segment are inhibited, the thalamic neurons that project to motor regions of the cortex are then released from the inhibition normally imposed on them by the medial pallidal segment. Because the projections from the thalamus to cortex are direct and excitatory, movement is then facilitated by the actions of the thalamocortical projection, which excites motor regions of cortex and their descending pathways to the brainstem and spinal cord. Thus, this feedback circuit can be interpreted to mean that movement follows activation of the neostriatum by the cortex. The functional relationships of the overall circuit for the direct pathway could now be summarized as follows, with (+) indicating excitation and (-) indicating inhibition (Fig. 20-8A): cerebral cortex (glutamate) → (+) neostriatum (GABA) → (-) internal pallidal segment (GABA) → (-) thalamus → (+) the motor regions of cortex. The net effect is excitation of the motor regions of cortex.In contrast, activation of the indirect pathway has a different effect on cortical neurons. The indirect pathway involves the following elements: (1) a GABAergic projection from the neostriatum to the external pallidal segment; (2) a GABAergic projection from the external pallidal segment to the subthalamic nucleus; and (3) a glutamatergic projection from the subthalamic nucleus to the internal pallidal segment. In this circuit, cortical activation initially results in activation of a GABAergic pathway from the neostriatum to the external pallidal segment, which inhibits neurons in this region. Because the pathway from the external pallidal segment to the subthalamic nucleus is also GABAergic and inhibitory, activation of striatal neurons results in inhibition of the (inhibitory) output pathway from the external pallidal segment to the subthalamic nucleus. As a consequence of activation of this circuit, the subthalamic nucleus is released from inhibition and can then provide a heightened glutamate-mediated excitatory input to the internal pallidal segment. Activation of the internal pallidal segment results in inhibition (via a GABAergic pathway) of the thalamic nuclei to which the neurons of the internal pallidal segment project. Thus, when this circuit is stimulated, the overall effect is to cause thalamic relay neurons to be inhibited, resulting in the reduction of excitation to the motor regions of cortex. Therefore, activation of the indirect pathway results in just the opposite effect (i.e., inhibition) on cortical motor neurons compared with the direct pathway. The functional relationships of the indirect pathway can now be summarized as follows, with (+) indicating excitation and (-) indicating inhibition: cerebral cortex (glutamate) → (+) neostriatum (GABA) → (-) external pallidal segment (GABA) → (-) subthalamic nucleus (glutamate) → (+) internal pallidal segment (GABA) → (-) thalamus → (+) motor regions of cortex. The net effect is inhibition of the motor regions of the cortex.
- The figure highlights how dopamine (DA) acting in the neostriatum (caudate nucleus–putamen) influences the activity of the direct and indirect pathways (black and turquoise arrows, respectively). Both pathways converge to regulate the activity of thalamocortical neurons. The activation of D1 dopamine receptors stimulates striatal output neurons of the direct pathway, leading to inhibition of GABA (-aminobutyric acid) neurons in the internal globus pallidus (GPi) and substantia nigra pars reticulata (SNr). In the indirect pathway, activation of D2 receptors inhibits striatal output neurons that project to the external globus pallidus (GPe). This results in the sequential inhibition of glutamatergic neurons in the subthalamic nucleus (STN) and GABA neurons in the GPi/SNr. The net effect of group I mGluRs is to counterbalance the action of dopamine across the direct pathway, although both mGluR1 and mGluR5 receptors stimulate GPe neurons. Activation of mGluR2/3 receptors mimics the action of dopamine by reducing glutamate release at corticostriatal and STN–GPi and STN–SNr synapses. mGluR4 receptor activation mimics the action of dopamine in the indirect pathway by inhibiting GABA release at the striatum–GPe synapse. For further information see Refs 12, 117, 118, 145, 146, 147. ACh, acetylcholine; CM thalamus, thalamic centromedian nucleus; Glut, glutamate; VA, VL, VM thalamus, ventral anterior, ventrolateral and ventromedial thalamic nuclei.
- (B) Hypokinetic disorder (Parkinson's disease). This model proposes that the hypokinetic effect is manifested by the reduced quantities of dopamine released, which act on dopamine D1 and D2 receptors in the neostriatum. Dopamine acting through D1 receptors in the neostriatum is excitatory to GABAergic neurons, which project to the GPM (direct pathway). Moreover, dopamine acting through D2 receptors in the neostriatum inhibits GABAergic neurons, which project to the GPL segment (indirect pathway). When there is diminished dopamine in the neostriatum released onto D1 receptors (dotted line), the resulting inhibitory output of the neostriatum on the GPM is diminished. This allows for an enhancement of the inhibitory output of the GPM to the thalamus to be increased (thicker arrow). Because the thalamus normally excites the cortex, greater inhibition on the thalamic nuclei by the GPM will cause a weakened input onto motor regions of the cortex. Likewise, the projection from the neostriatum, acting through D2 receptors, is inhibitory to the GPL. In turn, the GPL is normally inhibitory to the Sth. Thus, the diminished inhibitory input into the GPL from the neostriatum allows the GPL to exert greater inhibition on the Sth. Because the Sth normally excites the GPM via a glutamatergic mechanism, the reduced amount of inhibition on the Sth would allow it to exert a greater excitatory effect on the GPM. Therefore, the combined effects of reduced dopaminergic inputs into the neostriatum affect both the direct and indirect pathways in such a way as to allow for enhancement of the inhibitory outputs of the GPM on the thalamic projection nuclei and their target regions in motor cortex, causing the hypokinetic movement disorder.
- (C) Hyperkinetic disorder (Huntington's disease). Inhibitory influences mediated from the caudate and putamen to the external segment of the globus pallidus are diminished. Because of a reduction in GABA in the neostriatum, tonic inhibition mediated from the GPL to the Sth is enhanced, thus reducing the excitation from the Sth to the GPM. The diminished excitatory input to the GPM results in increased excitation to the VA and VL thalamic nuclei as well as the cortical receiving areas of these thalamic nuclei. The overall effect mediated on the motor regions of the cerebral cortex is an increase in excitation and greater (inappropriate) motor activity.
- SummaryThe contribution of the basal ganglia to motor control is apparent from the deficits that result from damage to the component nuclei. Such lesions compromise the initiation and performance of voluntary movements, as exemplified by the paucity of movement in Parkinson's disease and in the inappropriate "release" of movements in Huntington's disease. The organization of the basic circuitry of the basal ganglia indicates how this constellation of nuclei modulates movement. With respect to motor function, the system forms a loop that originates in almost every area of the cerebral cortex and eventually terminates, after enormous convergence within the basal ganglia, on the upper motor neurons in the motor and premotor areas of the frontal lobe and the superior colliculus. The efferent neurons of the basal ganglia influence the upper motor neurons in the cortex by gating the flow of information through relays in the ventral nuclei of the thalamus. The upper motor neurons in the superior colliculus that initiate saccadic eye movements are controlled by monosynaptic projections from substantia nigra pars reticulata. In each case, the basal ganglia loop regulates movement by a process of disinhibition that results from the serial interaction within the basal ganglia circuitry of two GABAergic neurons. Internal circuits within the basal ganglia system modulate the amplification of the signals that are transmitted through the loop.
