Mais conteúdo relacionado
Semelhante a Our Experiences by 2016 (20)
Our Experiences by 2016
- 2. Oncology Experience
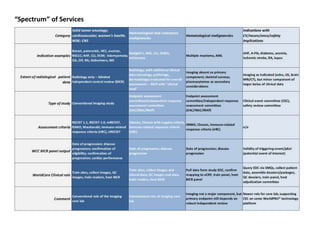
Indication Imaging Modalities
# ofStudies
# ofPatientsPh I Ph II Ph III Ph IV
BladderCancer CT, MRI, XR, Bone 2 1 1,100
Breastcancer CT, MRI, XR 1 4 7 3,600
ChronicLymphocyticLeukemia CT, MRI, PET 2 2 4 1,500
Colon CT, MRI, PET 3 1 2,200
Colorectal CT, MRI, PET 3 2 700
Diffuse Large B-Cell Lymphoma CT, MRI, PET 2 1 500
Esophagus CT, MRI, PET 1 200
FollicularLymphoma CT, MRI, PET 2 700
Gastric Cancer CT 1 3 2,300
HCC CT, MRI 3 1 800
Head& Neck CT, MRI, PET 3 1 340
Mantle Cell Lymphoma CT, MRI, PET 1 2 320
Melanoma CT, MRI, PET 2 3 4 3,800
Multiple Myeloma MRI 1 70
Non-HodgkinsLymphoma CT, MRI, PET 1 6 7 3,900
Non-Small Cell LungCancer CT, MRI, XR, Bone 3 8 3,600
Ovarian CT, MRI 3 4 2,600
Pancreatic CT, MRI, XR 1 3 1 1,400
Prostate Cancer CT, MRI, XR, Bone 1 5 6 7,000
RCC MRI, PET 1 1 900
Sarcoma CT, MRI 1 1 500
SolidTumors CT, MRI 2 75
- 3. Other Therapeutic Areas
CNS Phases # ofsites # ofpatients Modalities
Multiple Sclerosis II,III 40-160 10-1,500 MR
SubarachnoidHemorrhage III 100-150 750-1,500 CT, DSA,CTA
MSK Phases # ofsites # ofpatients Modalities
Osteoporosis I, II,III 1-400 12-17,000 X-Ray,DXA
RheumatoidArthritis I, II,III 1-80 10-1,500 X-Ray,MRI
Osteoarthritis I, II,III 1-350 12-2,250 X-Ray,MRI
Cardiology Phases # ofsites # ofpatients Modalities
Peripheral Arterial Occlusion III 90 100 Arteriograms
Fabry Disease II,III 2 60 Echo
Heart Failure III 40 600 Echo
Atrial Fibrillation Pivotal Device 25 220 CTA,TEE, CT, MR
EisenmengerSyndrome III 90 220 PA,CTPA,MRA, Echo
Pulmonary Phases # ofsites # ofpatients Modalities
Arterial Hypertension IV 30 100 PET, cMRI, 3D Echo
ChronicThromboembolicHypertension II 60 150 PA,CTPA,MRA, V/Q
Imaging Agents Phases # ofsites # ofpatients Modalities
Vascular III 15-25 35-300 MRA, DSA
Renal III 25 330 MRI
Oncology I, III 1 30-140 CT, PET-CT,SPECT-CT
- 4. Immunotherapy Experience
Phase Indication Agent Type Criteria Endpoint # of
sites
# of
subjects
III NSCLC Vaccine therapy RECIST 1.1 ProgressionFree Survival
(PFS)
89 506
III Non-Hodgkin’s
Lymphoma
Autologoustumorderived
immunoglobulinidiotypevaccine
Cheson Disease Free Survival (DFS) 30 650
III MetastaticMelanoma Oncolyticviral vaccine therapy ModifiedirRC Durable Response Rate 75 400
II MetastaticMelanoma Monoclonal antibody irRC & RECIST
1.1
PFS 31 100
I Hepatocellular
Carcinoma
Intralesionalinjection RECIST 1.1 PFS 2 45
III Colon Autologoustumorcell
immunotherapy
RECIST 1.1 DFS 40 550
II Ovarian Autologoustumorcell
immunotherapy
RECIST 1.1 Recurrence-FreeSurvival 80 400
III LocallyAdvanced
CutaneousMelanoma
Intralesionalinjection RECIST 1.1 PFS 30 225
III MetastaticMelanoma Oncolyticviral vaccine
therapy/anti-PD-1monoclonal
antibody
Modified
irRECIST&
RECIST
PFS 150 800