Properties of acids; bases
•
5 gostaram•921 visualizações
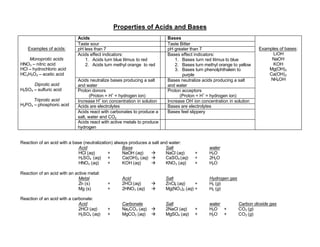
Acids have a sour taste, are electrolytes, turn indicators red, and have a pH less than 7. They donate protons and can neutralize bases to form salts and water. Bases have a bitter taste, are electrolytes, turn indicators blue or yellow, and have a pH greater than 7. They accept protons and can neutralize acids to form salts and water. Common acids include nitric acid, hydrochloric acid, acetic acid, sulfuric acid, and phosphoric acid. Common bases include lithium hydroxide, sodium hydroxide, potassium hydroxide, magnesium hydroxide, and calcium hydroxide.
Denunciar
Compartilhar
Denunciar
Compartilhar
Baixar para ler offline
Mais conteúdo relacionado
Mais procurados
Mais procurados (17)
Destaque
Destaque (15)
Semelhante a Properties of acids; bases
Semelhante a Properties of acids; bases (20)
Acids Bases Taste Taste pH greater than pH less tharn Acids effect ind.docx

Acids Bases Taste Taste pH greater than pH less tharn Acids effect ind.docx
Chemistry GCSE Chapter 8 Acid bases and Salts .pptx

Chemistry GCSE Chapter 8 Acid bases and Salts .pptx
PPT on Acids, Bases & Salts submitted by Bhavneet Singh.ppt

PPT on Acids, Bases & Salts submitted by Bhavneet Singh.ppt
Mais de Timothy Welsh
Mais de Timothy Welsh (20)
Último
TỔNG ÔN TẬP THI VÀO LỚP 10 MÔN TIẾNG ANH NĂM HỌC 2023 - 2024 CÓ ĐÁP ÁN (NGỮ Â...

TỔNG ÔN TẬP THI VÀO LỚP 10 MÔN TIẾNG ANH NĂM HỌC 2023 - 2024 CÓ ĐÁP ÁN (NGỮ Â...Nguyen Thanh Tu Collection
Último (20)
This PowerPoint helps students to consider the concept of infinity.

This PowerPoint helps students to consider the concept of infinity.
Interdisciplinary_Insights_Data_Collection_Methods.pptx

Interdisciplinary_Insights_Data_Collection_Methods.pptx
Kodo Millet PPT made by Ghanshyam bairwa college of Agriculture kumher bhara...

Kodo Millet PPT made by Ghanshyam bairwa college of Agriculture kumher bhara...
HMCS Max Bernays Pre-Deployment Brief (May 2024).pptx

HMCS Max Bernays Pre-Deployment Brief (May 2024).pptx
Micro-Scholarship, What it is, How can it help me.pdf

Micro-Scholarship, What it is, How can it help me.pdf
Beyond_Borders_Understanding_Anime_and_Manga_Fandom_A_Comprehensive_Audience_...

Beyond_Borders_Understanding_Anime_and_Manga_Fandom_A_Comprehensive_Audience_...
Food safety_Challenges food safety laboratories_.pdf

Food safety_Challenges food safety laboratories_.pdf
TỔNG ÔN TẬP THI VÀO LỚP 10 MÔN TIẾNG ANH NĂM HỌC 2023 - 2024 CÓ ĐÁP ÁN (NGỮ Â...

TỔNG ÔN TẬP THI VÀO LỚP 10 MÔN TIẾNG ANH NĂM HỌC 2023 - 2024 CÓ ĐÁP ÁN (NGỮ Â...
Jual Obat Aborsi Hongkong ( Asli No.1 ) 085657271886 Obat Penggugur Kandungan...

Jual Obat Aborsi Hongkong ( Asli No.1 ) 085657271886 Obat Penggugur Kandungan...
ICT role in 21st century education and it's challenges.

ICT role in 21st century education and it's challenges.
Properties of acids; bases
- 1. Properties of Acids and Bases Acids Bases Taste sour Taste Bitter pH less than 7 pH greater than 7 Acids effect indicators: 1. Acids turn blue litmus to red 2. Acids turn methyl orange to red Bases effect indicators: 1. Bases turn red litmus to blue 2. Bases turn methyl orange to yellow 3. Bases turn phenolphthalein to purple Acids neutralize bases producing a salt and water Bases neutralize acids producing a salt and water Proton donors (Proton = H+ = hydrogen ion) Proton acceptors (Proton = H+ = hydrogen ion) Increase H+ ion concentration in solution Increase OH- ion concentration in solution Acids are electrolytes Bases are electrolytes Acids react with carbonates to produce a salt, water and CO2 Bases feel slippery Examples of acids: Monoprotic acids HNO3 – nitric acid HCl – hydrochloric acid HC2H3O2 – acetic acid Diprotic acid H2SO4 – sulfuric acid Triprotic acid H3PO4 – phosphoric acid Acids react with active metals to produce hydrogen Examples of bases: LiOH NaOH KOH Mg(OH)2 Ca(OH)2 NH4OH Reaction of an acid with a base (neutralization) always produces a salt and water: Acid Base Salt water HCl (aq) + NaOH (aq) à NaCl (aq) + H2O H2SO4 (aq) + Ca(OH)2 (aq) à CaSO4 (aq) + 2H2O HNO3 (aq) + KOH (aq) à KNO3 (aq) + H2O Reaction of an acid with an active metal: Metal Acid Salt Hydrogen gas Zn (s) + 2HCl (aq) à ZnCl2 (aq) + H2 (g) Mg (s) + 2HNO3 (aq) à Mg(NO3)2 (aq) + H2 (g) Reaction of an acid with a carbonate: Acid Carbonate Salt water Carbon dioxide gas 2HCl (aq) + Na2CO3 (aq) à 2NaCl (aq) + H2O + CO2 (g) H2SO4 (aq) + MgCO3 (aq) à MgSO4 (aq) + H2O + CO2 (g)
