QPS Product Sheet Iron Sucrose
•
1 gostou•1,429 visualizações
QPS offers fully integrated front-to-end clinical and bioanalytical services for iron sucrose studies based on a vast track record.
Denunciar
Compartilhar
Denunciar
Compartilhar
Baixar para ler offline
Recomendados
Recomendados
Background and objectives: Iron is one of the major component
of hemoglobin, is required for transport of oxygen, ferritin is a
protein for storage iron, and transferring is a protein for iron
transport, all these it may be change in breast cancer.
The aim of present study was to measure the serum ferritin, iron,
total iron binding capacity, transferring, and C-reactive protein
concentration in breast tumors.
Material and method: A prospective study was carried out from
April 2013 to August 2014 by clinical biochemistry department in
College of Pharmacy-University of Sulaimani on (45) healthy
female individuals, (group 1) and (50) females with breast tumor
(group 2).
Results: The mean value of serum iron, transferring, total iron
binding capacity were significantly lower in females with breast
tumors (group 2), than that of healthy female individuals, (group
1), while serum ferritin was significantly higher in females with
breast tumors (group2), than that of healthy individuals (group
1).
Conclusion: Based on findings of the present study it can be
concluded that breast tumors can cause deficient of all iron
profile except ferritin will be increase in female breast cancers.
Key words- Serum iron, S.Ferritin, S. Total iron binding, S.
Transferrin, S.C-Reactive protein, breast tumors.FERRITIC AS A POTENT MARKER OF BREST CANCER

FERRITIC AS A POTENT MARKER OF BREST CANCERInternational Journal of Technical Research & Application
Mais conteúdo relacionado
Semelhante a QPS Product Sheet Iron Sucrose
Background and objectives: Iron is one of the major component
of hemoglobin, is required for transport of oxygen, ferritin is a
protein for storage iron, and transferring is a protein for iron
transport, all these it may be change in breast cancer.
The aim of present study was to measure the serum ferritin, iron,
total iron binding capacity, transferring, and C-reactive protein
concentration in breast tumors.
Material and method: A prospective study was carried out from
April 2013 to August 2014 by clinical biochemistry department in
College of Pharmacy-University of Sulaimani on (45) healthy
female individuals, (group 1) and (50) females with breast tumor
(group 2).
Results: The mean value of serum iron, transferring, total iron
binding capacity were significantly lower in females with breast
tumors (group 2), than that of healthy female individuals, (group
1), while serum ferritin was significantly higher in females with
breast tumors (group2), than that of healthy individuals (group
1).
Conclusion: Based on findings of the present study it can be
concluded that breast tumors can cause deficient of all iron
profile except ferritin will be increase in female breast cancers.
Key words- Serum iron, S.Ferritin, S. Total iron binding, S.
Transferrin, S.C-Reactive protein, breast tumors.FERRITIC AS A POTENT MARKER OF BREST CANCER

FERRITIC AS A POTENT MARKER OF BREST CANCERInternational Journal of Technical Research & Application
Semelhante a QPS Product Sheet Iron Sucrose (20)
Red Blood Cell Lipids, Serum Trace Elements and Immunological Markers in Pros...

Red Blood Cell Lipids, Serum Trace Elements and Immunological Markers in Pros...
IDENTIFYING PARATHYROID HORMONE DISORDERS AND ITS PHENOTYPES THROUGH A BONE H...

IDENTIFYING PARATHYROID HORMONE DISORDERS AND ITS PHENOTYPES THROUGH A BONE H...
Identifying metabolic markers of pre-obesity regulated by growth hormone usin...

Identifying metabolic markers of pre-obesity regulated by growth hormone usin...
A Systematic Review of the Cobalt Content of the Normal Human Prostate Gland

A Systematic Review of the Cobalt Content of the Normal Human Prostate Gland
Analysis of Body Fluids using Mass spectroscopy.pptx

Analysis of Body Fluids using Mass spectroscopy.pptx
External quality assurance in clinical trace element labs 

External quality assurance in clinical trace element labs
Mais de QPS Holdings, LLC
Mais de QPS Holdings, LLC (20)
QPS Regulated Bioanalysis of Antibody Drug Conjugates

QPS Regulated Bioanalysis of Antibody Drug Conjugates
Three Lessons to Help Accelerate Pharmaceutical Breakthroughs for CNS Disorders

Three Lessons to Help Accelerate Pharmaceutical Breakthroughs for CNS Disorders
Último
Último (20)
💚Reliable Call Girls Chandigarh 💯Niamh 📲🔝8868886958🔝Call Girl In Chandigarh N...

💚Reliable Call Girls Chandigarh 💯Niamh 📲🔝8868886958🔝Call Girl In Chandigarh N...
Gorgeous Call Girls Dehradun {8854095900} ❤️VVIP ROCKY Call Girls in Dehradun...

Gorgeous Call Girls Dehradun {8854095900} ❤️VVIP ROCKY Call Girls in Dehradun...
Chennai ❣️ Call Girl 6378878445 Call Girls in Chennai Escort service book now

Chennai ❣️ Call Girl 6378878445 Call Girls in Chennai Escort service book now
❤️Amritsar Escorts Service☎️9815674956☎️ Call Girl service in Amritsar☎️ Amri...

❤️Amritsar Escorts Service☎️9815674956☎️ Call Girl service in Amritsar☎️ Amri...
Call Girls Shahdol Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Shahdol Just Call 8250077686 Top Class Call Girl Service Available
💰Call Girl In Bangalore☎️7304373326💰 Call Girl service in Bangalore☎️Bangalor...

💰Call Girl In Bangalore☎️7304373326💰 Call Girl service in Bangalore☎️Bangalor...
Kolkata Call Girls Naktala 💯Call Us 🔝 8005736733 🔝 💃 Top Class Call Girl Se...

Kolkata Call Girls Naktala 💯Call Us 🔝 8005736733 🔝 💃 Top Class Call Girl Se...
Cheap Rate Call Girls Bangalore {9179660964} ❤️VVIP BEBO Call Girls in Bangal...

Cheap Rate Call Girls Bangalore {9179660964} ❤️VVIP BEBO Call Girls in Bangal...
Exclusive Call Girls Bangalore {7304373326} ❤️VVIP POOJA Call Girls in Bangal...

Exclusive Call Girls Bangalore {7304373326} ❤️VVIP POOJA Call Girls in Bangal...
Circulatory Shock, types and stages, compensatory mechanisms

Circulatory Shock, types and stages, compensatory mechanisms
Gastric Cancer: Сlinical Implementation of Artificial Intelligence, Synergeti...

Gastric Cancer: Сlinical Implementation of Artificial Intelligence, Synergeti...
👉 Chennai Sexy Aunty’s WhatsApp Number 👉📞 7427069034 👉📞 Just📲 Call Ruhi Colle...

👉 Chennai Sexy Aunty’s WhatsApp Number 👉📞 7427069034 👉📞 Just📲 Call Ruhi Colle...
💚Chandigarh Call Girls Service 💯Piya 📲🔝8868886958🔝Call Girls In Chandigarh No...

💚Chandigarh Call Girls Service 💯Piya 📲🔝8868886958🔝Call Girls In Chandigarh No...
Chandigarh Call Girls Service ❤️🍑 9809698092 👄🫦Independent Escort Service Cha...

Chandigarh Call Girls Service ❤️🍑 9809698092 👄🫦Independent Escort Service Cha...
Pune Call Girl Service 📞9xx000xx09📞Just Call Divya📲 Call Girl In Pune No💰Adva...

Pune Call Girl Service 📞9xx000xx09📞Just Call Divya📲 Call Girl In Pune No💰Adva...
Chandigarh Call Girls Service ❤️🍑 9809698092 👄🫦Independent Escort Service Cha...

Chandigarh Call Girls Service ❤️🍑 9809698092 👄🫦Independent Escort Service Cha...
❤️Chandigarh Escorts Service☎️9814379184☎️ Call Girl service in Chandigarh☎️ ...

❤️Chandigarh Escorts Service☎️9814379184☎️ Call Girl service in Chandigarh☎️ ...
Call Girls Bangalore - 450+ Call Girl Cash Payment 💯Call Us 🔝 6378878445 🔝 💃 ...

Call Girls Bangalore - 450+ Call Girl Cash Payment 💯Call Us 🔝 6378878445 🔝 💃 ...
❤️Call Girl Service In Chandigarh☎️9814379184☎️ Call Girl in Chandigarh☎️ Cha...

❤️Call Girl Service In Chandigarh☎️9814379184☎️ Call Girl in Chandigarh☎️ Cha...
QPS Product Sheet Iron Sucrose
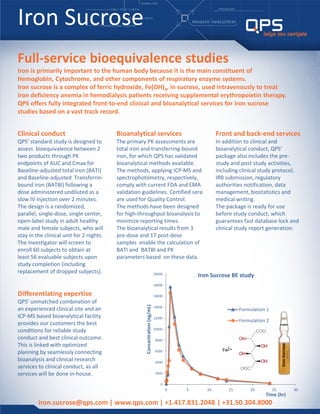
- 1. Iron Sucrose iron.sucrose@qps.com | www.qps.com | +1.417.831.2048 | +31.50.304.8000 Full-service bioequivalence studies Iron is primarily important to the human body because it is the main constituent of Hemoglobin, Cytochrome, and other components of respiratory enzyme systems. Iron sucrose is a complex of ferric hydroxide, Fe(OH)3, in sucrose, used intravenously to treat iron deficiency anemia in hemodialysis patients receiving supplemental erythropoietin therapy. QPS offers fully integrated front-to-end clinical and bioanalytical services for iron sucrose studies based on a vast track record. Clinical conduct QPS’ standard study is designed to assess bioequivalence between 2 two products through PK endpoints of AUC and Cmax for Baseline-adjusted total iron (BATI) and Baseline-adjusted Transferrin- bound iron (BATBI) following a dose administered undiluted as a slow IV injection over 2 minutes. The design is a randomized, parallel, single-dose, single center, open-label study in adult healthy male and female subjects, who will stay in the clinical unit for 2 nights. The investigator will screen to enroll 60 subjects to obtain at least 56 evaluable subjects upon study completion (including replacement of dropped subjects). Front and back-end services In addition to clinical and bioanalytical conduct, QPS’ package also includes the pre- study and post study activities, including clinical study protocol, IRB submission, regulatory authorities notification, data management, biostatistics and medical writing. The package is ready for use before study conduct, which guarantees fast database lock and clinical study report generation. Bioanalytical services The primary PK assessments are total iron and transferring-bound iron, for which QPS has validated bioanalytical methods available. The methods, applying ICP-MS and spectrophotometry, respectively, comply with current FDA and EMA validation guidelines. Certified sera are used for Quality Control. The methods have been designed for high-throughput bioanalysis to minimize reporting times. The bioanalytical results from 3 pre-dose and 17 post-dose samples enable the calculation of BATI and BATBI and PK parameters based on these data. Differentiating expertise QPS’ unmatched combination of an experienced clinical site and an ICP-MS based bioanalytical facility provides our customers the best conditions for reliable study conduct and best clinical outcome. This is linked with optimized planning by seamlessly connecting bioanalysis and clinical research services to clinical conduct, as all services will be done in-house. 0 2000 4000 6000 8000 10000 12000 14000 16000 18000 20000 0 5 10 15 20 25 30 Concentration(ng/mL) Time (hr) Iron Sucrose BE study Formulation 1 Formulation 2
