Final Review
•Transferir como PPT, PDF•
2 gostaram•828 visualizações
Denunciar
Compartilhar
Denunciar
Compartilhar
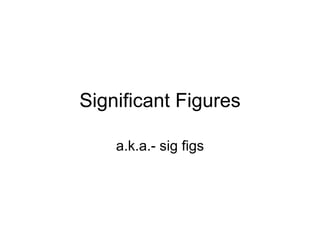
Recomendados
Recomendados
Mais conteúdo relacionado
Mais procurados
Mais procurados (20)
IA on effectiveness of different types of catalysts MnO2 vs Fe(NO3)3 on the r...

IA on effectiveness of different types of catalysts MnO2 vs Fe(NO3)3 on the r...
Temperature and Kinetic Theory of Gases cheat sheet

Temperature and Kinetic Theory of Gases cheat sheet
IA on effect of different transition metal (homogenous catalyst) on the decom...

IA on effect of different transition metal (homogenous catalyst) on the decom...
Destaque (7)
Camera shots and angles for a horror and thriller film

Camera shots and angles for a horror and thriller film
Semelhante a Final Review
Semelhante a Final Review (20)
Notes for Unit 17 of AP Chemistry (Thermodynamics)

Notes for Unit 17 of AP Chemistry (Thermodynamics)
Mais de Frederick High School
Mais de Frederick High School (20)
Último
Último (20)
Asian American Pacific Islander Month DDSD 2024.pptx

Asian American Pacific Islander Month DDSD 2024.pptx
On National Teacher Day, meet the 2024-25 Kenan Fellows

On National Teacher Day, meet the 2024-25 Kenan Fellows
Beyond the EU: DORA and NIS 2 Directive's Global Impact

Beyond the EU: DORA and NIS 2 Directive's Global Impact
Food Chain and Food Web (Ecosystem) EVS, B. Pharmacy 1st Year, Sem-II

Food Chain and Food Web (Ecosystem) EVS, B. Pharmacy 1st Year, Sem-II
Role Of Transgenic Animal In Target Validation-1.pptx

Role Of Transgenic Animal In Target Validation-1.pptx
Measures of Central Tendency: Mean, Median and Mode

Measures of Central Tendency: Mean, Median and Mode
Measures of Dispersion and Variability: Range, QD, AD and SD

Measures of Dispersion and Variability: Range, QD, AD and SD
Basic Civil Engineering first year Notes- Chapter 4 Building.pptx

Basic Civil Engineering first year Notes- Chapter 4 Building.pptx
Unit-V; Pricing (Pharma Marketing Management).pptx

Unit-V; Pricing (Pharma Marketing Management).pptx
Russian Escort Service in Delhi 11k Hotel Foreigner Russian Call Girls in Delhi

Russian Escort Service in Delhi 11k Hotel Foreigner Russian Call Girls in Delhi
Final Review
- 1. Significant Figures a.k.a.- sig figs
- 9. + - - - NaCl Na = Cl = H 2 O H = O = + - + Process of Solvation - + + - + + + - - + + - + - - + - + - + - + - + + - + + - + + - + + - + +
- 20. Alpha Decay Example Notice that the uranium has changed into a new element, thorium.
- 22. Beta Decay Example Notice that carbon has changed into nitrogen.
- 24. Gamma Decay Example No new element formed. Gamma radiation (energy) released.
- 27. Average Atomic Mass (How the number ends up on the periodic table!!) 1 st Mass of one isotope x % abundance in decimal form (watch SIG FIGS!!) 2 nd Do this for each isotope of that element 3 rd Add all individual isotopes together to get the average atomic mass of the element.
- 28. 1. Calculate the average atomic mass of potassium using the following data: Potassium-39 38.964 amu x 0.9312 = 36.28 amu Potassium-41 40.962 amu x 0.0688 2.82 amu = + Average atomic mass for K = 39.10 amu 6.88 % 40.962 amu Potassium-41 93.12% 38.964 amu Potassium-39 % abundance Mass Isotope
- 29. 2. Calculate the average atomic mass of magnesium using the following data: Magnesium-24 23.985 amu x 0.7870 = 18.88 amu Magnesium-25 24.986 amu x 0.1013 2.531 amu = + Average atomic mass for K = 24.31 amu + Magnesium-26 25.983 amu x 0.1117 = 2.902 amu 11.17 % 25.983 amu Magnesium-26 10.13 % 24.986 amu Magnesium-25 78.70% 23.985 amu Magnesium-24 % abundance Mass Isotope