Casey Clinical Summary | ChloraPrep UK Infection Prevention & Control
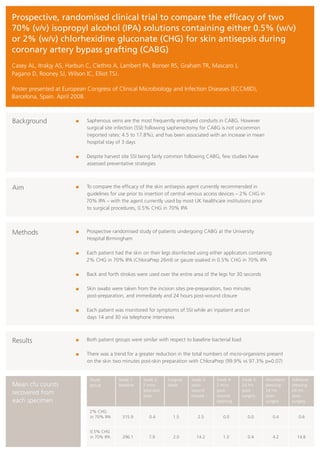
- 1. Prospective, randomised clinical trial to compare the efficacy of two 70% (v/v) isopropyl alcohol (IPA) solutions containing either 0.5% (w/v) or 2% (w/v) chlorhexidine gluconate (CHG) for skin antisepsis during coronary artery bypass grafting (CABG) Casey AL, Itrakjy AS, Harbun C, Clethro A, Lambert PA, Bonser RS, Graham TR, Mascaro J, Pagano D, Rooney SJ, Wilson IC, Elliot TSJ. Poster presented at European Congress of Clinical Microbiology and Infection Diseases (ECCMID), Barcelona, Spain. April 2008. Background Saphenous veins are the most frequently employed conduits in CABG. However surgical site infection (SSI) following saphenectomy for CABG is not uncommon (reported rates: 4.5 to 17.8%), and has been associated with an increase in mean hospital stay of 3 days Despite harvest site SSI being fairly common following CABG, few studies have assessed preventative strategies Aim To compare the efficacy of the skin antisepsis agent currently recommended in guidelines for use prior to insertion of central venous access devices – 2% CHG in 70% IPA – with the agent currently used by most UK healthcare institutions prior to surgical procedures, 0.5% CHG in 70% IPA Methods Prospective randomised study of patients undergoing CABG at the University Hospital Birmingham Each patient had the skin on their legs disinfected using either applicators containing 2% CHG in 70% IPA (ChloraPrep 26ml) or gauze soaked in 0.5% CHG in 70% IPA Back and forth strokes were used over the entire area of the legs for 30 seconds Skin swabs were taken from the incision sites pre-preparation, two minutes post-preparation, and immediately and 24 hours post-wound closure Each patient was monitored for symptoms of SSI while an inpatient and on days 14 and 30 via telephone interviews Results Both patient groups were similar with respect to baseline bacterial load There was a trend for a greater reduction in the total numbers of micro-organisms present on the skin two minutes post-skin preparation with ChloraPrep (99.9% vs 97.3% p=0.07) Study Swab 1: Swab 2: Surgical Swab 3: Swab 4: Swab 5: Absorbent Adhesive Mean cfu counts group baseline 2 mins blade post- 2 mins 24 hrs dressing: dressing: post-skin wound post- post- 24 hrs 24 hrs recovered from prep closure wound surgery post- post- each specimen cleaning surgery surgery 2% CHG in 70% IPA 315.9 0.4 1.5 2.5 0.0 0.0 0.4 0.6 0.5% CHG in 70% IPA 296.1 7.9 2.0 14.2 1.3 0.4 4.2 14.8
- 2. With ChloraPrep, both the absorbent and adhesive components of the dressings removed 24 hours post-surgery contained a significantly lower number of micro-organisms than those in the 0.5% CHG/70% IPA group (p=0.02 and p=0.007 respectively) Fewer ChloraPrep patients developed a superficial SSI post-discharge than 0.5% CHG patients (p=0.0502) 25 p=0.49 p=0.0502 Incidence of superficial SSI 20 post-discharge 20.8 (% patients) 15 10 5 0 0 0.5% chlorhexidine/ ChloraPrep 70% isopropyl alcohol n=22 n=24 Conclusions The results of this interim analysis suggest that for microbial reduction two minutes post-application, ChloraPrep outperforms 0.5% CHG in 70% IPA The reduced number of micro-organisms in the dressings removed from ChloraPrep patients may reflect superior residual activity with this antiseptic While more patients need to be evaluated, ChloraPrep has the potential to reduce the risk of SSI Prescribing Information contact with eyes, mucous membranes, middle ear and neural tissue. Should not be used in ChloraPrep® (PL31760/0002) & ChloraPrep with Tint (PL31760-0001) 2% chlorhexidine children under 2 months of age. Solution is flammable. Do not use with ignition sources until gluconate w/v / 70% isopropyl alcohol v/v cutaneous solution. Indication: Disinfection of dry, do not allow to pool, and remove soaked materials before use. Over-vigorous use on skin prior to invasive medical procedures. Dosage & administration: ChloraPrep – 0.67ml, fragile or sensitive skin or repeated use may lead to local skin reactions. At the first sign of 1.5ml, 3ml, 10.5ml, 26ml; ChloraPrep with Tint – 3ml, 10.5ml, 26ml. Volume dependent on local skin reaction, application should be stopped. Per applicator costs (ex VAT) ChloraPrep invasive procedure being undertaken. Applicator squeezed to break ampoule and release – 0.67ml (SEPP) - 30p; 1.5ml (FREPP) - 55p; 3ml - 85p; 10.5ml - £2.92; 26ml - £6.50. antiseptic solution onto sponge. Solution applied by gently pressing sponge against skin and ChloraPrep with Tint – 3ml - 89p; 10.5ml - £3.07; 26ml - £6.83. Legal category: GSL. moving back and forth for 30 seconds. The area covered should be allowed to air dry. Side Marketing Authorisation Holder: CareFusion UK 244 Ltd, 43 London Road, Reigate, Surrey effects, precautions & contra-indications: Very rarely allergic or skin reactions reported RH2 9PW, UK. Date of preparation: July 2010. with chlorhexidine, isopropyl alcohol and Sunset Yellow. Contra-indicated for patients with known hypersensitivity to these constituents. For external use only on intact skin. Avoid CHL112a Date of preparation: August 2010
