Bunya virus- Bunyaviridae
•Transferir como PPTX, PDF•
4 gostaram•1,474 visualizações
Bunyavirus, any virus belonging to the family Bunyaviridae. Bunyaviridae is a family of arthropod-borne or rodent-borne, spherical, enveloped RNA viruses. Bunyaviruses are responsible for a number of febrile diseases in humans and other vertebrates. They have either a rodent host or an arthropod vector and a vertebrate host.
Denunciar
Compartilhar
Denunciar
Compartilhar
Recomendados
Mais conteúdo relacionado
Mais procurados
Mais procurados (20)
Orthomyxovirus - Morphology and laboratory diagnosis

Orthomyxovirus - Morphology and laboratory diagnosis
Semelhante a Bunya virus- Bunyaviridae
Semelhante a Bunya virus- Bunyaviridae (20)
Viral classification and Types of Replication in virus 

Viral classification and Types of Replication in virus
Mais de Anup Bajracharya
Mais de Anup Bajracharya (20)
Type II Hypersensitivity-Antibody mediated cytotoxic Hypersensitivity

Type II Hypersensitivity-Antibody mediated cytotoxic Hypersensitivity
Microbiology -T.U. B.Sc. 1st Year General Microbiology Syllabus (Nepal)

Microbiology -T.U. B.Sc. 1st Year General Microbiology Syllabus (Nepal)
Pentose phosphate pathway (Hexose Monophosphate Pathway)

Pentose phosphate pathway (Hexose Monophosphate Pathway)
Environmental impacts of coronavirus crisis, challenges ahead

Environmental impacts of coronavirus crisis, challenges ahead
Último
Último (20)
💚Chandigarh Call Girls 💯Riya 📲🔝8868886958🔝Call Girls In Chandigarh No💰Advance...

💚Chandigarh Call Girls 💯Riya 📲🔝8868886958🔝Call Girls In Chandigarh No💰Advance...
❤️Chandigarh Escorts Service☎️9814379184☎️ Call Girl service in Chandigarh☎️ ...

❤️Chandigarh Escorts Service☎️9814379184☎️ Call Girl service in Chandigarh☎️ ...
Kolkata Call Girls Naktala 💯Call Us 🔝 8005736733 🔝 💃 Top Class Call Girl Se...

Kolkata Call Girls Naktala 💯Call Us 🔝 8005736733 🔝 💃 Top Class Call Girl Se...
👉 Amritsar Call Girls 👉📞 8725944379 👉📞 Just📲 Call Ruhi Call Girl Near Me Amri...

👉 Amritsar Call Girls 👉📞 8725944379 👉📞 Just📲 Call Ruhi Call Girl Near Me Amri...
❤️Call Girl Service In Chandigarh☎️9814379184☎️ Call Girl in Chandigarh☎️ Cha...

❤️Call Girl Service In Chandigarh☎️9814379184☎️ Call Girl in Chandigarh☎️ Cha...
Nagpur Call Girl Service 📞9xx000xx09📞Just Call Divya📲 Call Girl In Nagpur No💰...

Nagpur Call Girl Service 📞9xx000xx09📞Just Call Divya📲 Call Girl In Nagpur No💰...
Jaipur Call Girl Service 📞9xx000xx09📞Just Call Divya📲 Call Girl In Jaipur No💰...

Jaipur Call Girl Service 📞9xx000xx09📞Just Call Divya📲 Call Girl In Jaipur No💰...
Call Girls Mussoorie Just Call 8854095900 Top Class Call Girl Service Available

Call Girls Mussoorie Just Call 8854095900 Top Class Call Girl Service Available
Circulatory Shock, types and stages, compensatory mechanisms

Circulatory Shock, types and stages, compensatory mechanisms
Call 8250092165 Patna Call Girls ₹4.5k Cash Payment With Room Delivery

Call 8250092165 Patna Call Girls ₹4.5k Cash Payment With Room Delivery
Gorgeous Call Girls Dehradun {8854095900} ❤️VVIP ROCKY Call Girls in Dehradun...

Gorgeous Call Girls Dehradun {8854095900} ❤️VVIP ROCKY Call Girls in Dehradun...
Chandigarh Call Girls Service ❤️🍑 9809698092 👄🫦Independent Escort Service Cha...

Chandigarh Call Girls Service ❤️🍑 9809698092 👄🫦Independent Escort Service Cha...
💚Chandigarh Call Girls Service 💯Piya 📲🔝8868886958🔝Call Girls In Chandigarh No...

💚Chandigarh Call Girls Service 💯Piya 📲🔝8868886958🔝Call Girls In Chandigarh No...
Jual Obat Aborsi Di Dubai UAE Wa 0838-4800-7379 Obat Penggugur Kandungan Cytotec

Jual Obat Aborsi Di Dubai UAE Wa 0838-4800-7379 Obat Penggugur Kandungan Cytotec
Chandigarh Call Girls Service ❤️🍑 9809698092 👄🫦Independent Escort Service Cha...

Chandigarh Call Girls Service ❤️🍑 9809698092 👄🫦Independent Escort Service Cha...
💰Call Girl In Bangalore☎️63788-78445💰 Call Girl service in Bangalore☎️Bangalo...

💰Call Girl In Bangalore☎️63788-78445💰 Call Girl service in Bangalore☎️Bangalo...
Call Girl In Indore 📞9235973566📞 Just📲 Call Inaaya Indore Call Girls Service ...

Call Girl In Indore 📞9235973566📞 Just📲 Call Inaaya Indore Call Girls Service ...
Bandra East [ best call girls in Mumbai Get 50% Off On VIP Escorts Service 90...

Bandra East [ best call girls in Mumbai Get 50% Off On VIP Escorts Service 90...
Bunya virus- Bunyaviridae
- 1. Bunya virus Anup Muni Bajracharya
- 2. Bunya virus • Bunyavirus, any virus belonging to the family Bunyaviridae. • a very large family • single-strand, enveloped RNA viruses (more than 300 viruses) • consists of five genera of viruses: • Orthobunyavirus, • Phlebovirus, • Nairovirus, • Hantavirus, and • Tospovirus (Tospoviruses infect only plants). • Most transmitted by arthropods (e.g., ticks, mosquitoes, and sand flies) • cause serious human disease, including certain types of viral hemorrhagic fever.
- 3. Bunya virus • The name Bunyavirales derives from Bunyamwera where the original type species Bunyamwera orthobunyavirus was first discovered. • Ellioviricetes is named in honor of late virologist Richard M. Elliott for his early work on bunyaviruses. • Bunyamwera is a town in Bundibugyo District, Uganda.
- 4. Four genera that infect animals • Genus Orthobunyavirus • contains a large number of viruses that share common genetic features and are serologically unrelated to viruses in other genera of the Bunyaviridae. • are mosquito-borne, but some are transmitted by sandflies or midges (Culicoides spp.). • includes a number of pathogens of domestic animals and humans, including Akabane and La Crosse viruses and their relatives. • Genus Phlebovirus • contains over 50 viruses, all of which are transmitted by sandflies or mosquitoes. • contains important pathogens, including Rift Valley fever virus and the sandfly fever viruses.
- 5. • Genus Nairovirus • contains a large number of viruses, most of which are tick-borne, including the pathogens Nairobi sheep disease and Crimean–Congo hemorrhagic fever viruses. • Genus Hantavirus • also includes a substantial number of viruses, • are transmitted by persistently infected reservoir rodent hosts via urine, feces, and saliva • In humans, several of these viruses from Asia cause hemorrhagic fever with renal syndrome, whereas those from Europe are typically associated with a different and less severe disease syndrome. • Some of the hantaviruses from the Americas cause a severe acute respiratory distress syndrome referred to as “hantavirus pulmonary syndrome.”
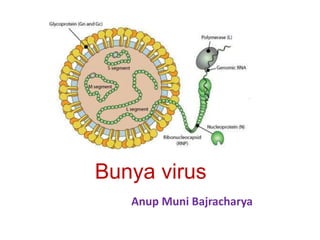
- 6. Figure -Diagrammatic representation of a bunyavirus virion (A) Gc, Gn, glycoproteins produced by processing of M RNA polyprotein; L, transcriptase encoded by L RNA; L, M, and S RNA, large, medium, and small RNA segments; N, nucleoprotein encoded by S RNA. (B) Hepatocyte of a rat infected with Rift Valley fever virus, showing virions budding in Golgi vesicles. (C) Thin section of mouse brain infected with California encephalitis virus, showing extracellular virions. (D) Negatively stained Hantaan virus virions, showing the pattern of spike placement in squares that is characteristic of all hantaviruses. (E) Negatively stained Rift Valley fever virus virions, showing the delicate spike fringe. Bars represent 100 nm.
- 7. Genome of Bunyaviruses • 11–19 kb • consists of three segments of negative-sense (or ambisense), single- stranded RNA, designated large (L), medium (M), and small (S), differ in size • the L RNA segment (6.3 to 12 kb), encodes a single large protein (L), the RNA-dependent RNA polymerase. • the M RNA segment (3.5 to 6 kb), encodes a polyprotein that is processed to form two glycoproteins (Gn and Gc) and, in some cases, a nonstructural protein (NSm). • the S RNA segment from 1 to 2.2 kb, encodes the nucleoprotein (N) and, for members of the Orthobunyavirus and Phlebovirus genera, a nonstructural (NSs) protein.
- 8. Virus Replication • Viral entry -by receptor-mediated endocytosis; all subsequent steps take place in the cytoplasm. • Cell receptors are not described for many bunyaviruses. • Exception -in hantaviruses, cell receptors include αβ integrins and other cell receptor proteins such as gC1qR/p32, which is expressed on endothelial cells, dendritic cells, lymphocytes, and platelets. • Because the genome of the single-stranded, negative- sense RNA ----viruses cannot be translated directly, the first step after penetration of the host cell and uncoating is the activation of the virion RNA polymerase (transcriptase) and its transcription of viral mRNAs from each of the three virion RNAs.
- 9. • The RNA polymerase also has endonuclease activity, cleaving 5′-methylated caps from host mRNAs and adding these to viral mRNAs to prime transcription (so-called cap snatching). • After primary viral mRNA transcription and translation, replication of the virion RNA occurs and a second round of transcription begins, with preferential amplification of the genes that encode structural proteins necessary for virion synthesis. • Virions mature by budding through intracytoplasmic vesicles associated with the Golgi complex and are released by the transport of vesicles through the cytoplasm and subsequent exocytosis from the apical and/or basolateral plasma membranes Virus Replication
- 10. Life Cycle
- 11. Pathogenesis • Except for members of the genus Hantavirus, bunyaviruses replicate in arthropods. • The gut of the vector is infected initially, and after a few days or weeks the virus appears in the saliva; the arthropod then remains infective for life but is not ill. • When the vector takes a blood meal, the infective saliva enters the small capillaries or lymphatics of the human or other vertebrate host. • The primary site of replication in humans is not known; it may be the vascular endothelium, the skin, or the regional lymph nodes. An incubation period of a few days ensues, after which the vertebrate host develops viremia. • Fever accompanies viremia, which seeds the liver in Rift Valley fever and Crimean- Congo hemorrhagic fever. Encephalitis, retinitis, and renal involvement usually appear later in infection (after antibody formation).
- 12. Laboratory Diagnosis Bunyavirus Infections • by isolating the virus, • detecting RNA by RT-PCR, or • by showing a fourfold or greater rise in antibody titer between acute- and convalescent-phase sera. • The virus can be isolated from blood (or from brain, liver, and other organs postmortem) during the viremic phase, but not usually after the third day of fever. • Virus Isolation - intracranial inoculation of suckling mice is thought to be the most sensitive system available for virus isolation. However, several sensitive cell culture systems are available such as vero. LLC-MC2 and mosquito cells. Once isolated the virus can be types by neutralizing tests. • Rapid Diagnosis - antigen detection systems and the detection of specific IgM antibodies are becoming available as means of rapid diagnosis. • Serology - a wide variety of serological techniques are available such as HI, CFT, IFA, neutralization tests and ELISAs.
- 13. Prevention • depends on the reservoir, amplifying hosts and how the viruses are transmitted, i.e. the vector, whether ticks or mosquitoes and which animals are involved. • general hygiene • limiting contact with vector saliva, urine, feces, or bedding. • No licensed vaccine for bunyaviruses.
- 14. References • https://www.sciencedirect.com/topics/neuroscience/b unyaviridae • https://www.ncbi.nlm.nih.gov/books/NBK8004/ • https://viralzone.expasy.org/6776 • https://www.britannica.com/science/bunyavirus • Textbook of Microbiology and Immunology- S Parija • The short textbook of Medical Microbiology-Gupte
