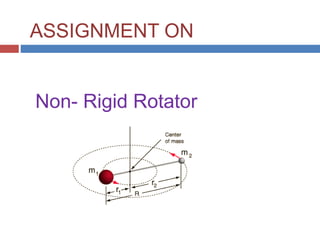
Non Rigid Rotator
- 1. ASSIGNMENT ON Non- Rigid Rotator
- 2. Rotational spectroscopy Rotational spectroscopy is concerned with the measurement of the energies of transitions between quantized rotational states of molecules in the gas phase. The spectra of polar molecules can be measured in absorption or emission by microwave spectroscopy or by far infrared spectroscopy. The rotational spectra of non-polar molecules cannot be observed by those methods, but can be observed and measured by Raman spectroscopy. Rotational spectroscopy is sometimes referred to as pure rotational spectroscopy to distinguish it from rotational-vibrational spectroscopy where changes in rotational energy occur together with changes in vibrational energy, and also from ro-vibronic spectroscopy where rotational, vibrational and electronic energy changes occur simultaneously
- 3. Types of molecule Linear molecule Symmetric top Molecule Spherical top molecule Asymmetric top Molecule
- 4. Rotational Spectra Simplest Case: Diatomic or Linear Polyatomic molecule Rigid Rotor Model: Two nuclei joined by a weightless rod J = Rotational quantum number (J = 0, 1, 2, …) I = Moment of inertia = mr2 m = reduced mass = m1m2 / (m1 + m2) r = internuclear distance m1 m2 1JJ I2 E 2 J
- 5. Rigid Rotor Model In wavenumbers (cm-1): 1JJ Ic8 h F 2J 1JJBFJ Separation between adjacent levels: F(J) – F(J-1) = 2BJ
- 6. Rotational Energy Levels Selection Rules: Molecule must have a permanent dipole. DJ = 1 J. Michael Hollas, Modern Spectroscopy, John Wiley & Sons, New York, 1992.
- 7. The Non-Rigid Rotor Account for the dynamic nature of the chemical bond: DJ = 0, 1 22 J 1)(JJ hc D 1JJ hc B E D is the centrifugal distortion constant (D is large when a bond is easily stretched) Typically, D < 10-4*B and B = 0.1 – 10 cm-1 22 J 1)(JJD1JJB F m k c D 2 1 and B4 2 3
- 8. Vibrational Transitions Simplest Case: Diatomic Molecule Harmonic Oscillator Model: Two atoms connected by a spring. 1/2E vv 1/2hE vv v = vibrational quantum number (v = 0, 1, 2, …) = classical vibrational frequency 2/1 k 2 1 m k = force constant (related to the bond order). in Joules in cm-1
- 9. Vibrational Energy Levels J. Michael Hollas, Modern Spectroscopy, John Wiley & Sons, New York, 1992. Selection Rules: 1) Must have a change in dipole moment (for IR). 2) Dv = 1
- 10. Anharmonicity Ingle and Crouch, Spectrochemical Analysis ...1/2-1/2E 2 e vvv Selection Rules: Dv = 1, 2, 3, … Dv = 2, 3, … are called overtones. Overtones are often weak because anharmonicity at low v is small.
- 11. Rotation – Vibration Transitions The rotational selection rule during a vibrational transition is: DJ = 1 Unless the molecule has an odd number of electrons (e.g. NO). Then, DJ = 0, 1 0,1,2..Jand0,1,2,...for11/2E v vJJBvvJ Bv signifies the dependence of B on vibrational level
- 12. Rotation – Vibration Transitions Ingle and Crouch, Spectrochemical Analysis If DJ = -1 P – Branch If DJ = 0 Q – Branch If DJ = +1 R – Branch
- 13. Thank you