What’s next: The future of non-invasive neurotechnology
- 1. What’s next: The future of non-invasive neurotechnology
- 3. What’s next: The future of non-invasive neurotechnology Chaired by: James Cavuoto, Editor and Publisher at Neurotech Reports Dr. Bruce Cuthbert Director of Adult Translational Research and Treatment Development at the NIMH Charles Fisher, President of Fisher Wallace Laboratories
- 4. What’s Next: The future of targets for non-invasive neurotechnology SharpBrains Virtual Summit October 29, 2014 Bruce N. Cuthbert, Ph. D. Director, NIMH RDoC Unit National Institute of Mental Health
- 5. Why RDoC? ~~ Problems with the DSM system for research …. • Current diagnostic systems (DSM/ICD) based on clinical symptoms and signs: cannot identify biological/psychological mechanisms of disorders. • Waiting until symptoms appear: makes prevention more difficult • Problems such as heterogeneity, co-morbidity, over-specification, NOS diagnoses • BUT: DSM/ICD is the de facto standard for research grants – as well as journal publications, clinical trials, and regulatory approval. 5
- 6. …. and problems for clinical use (& new treatments) 6 “On average, a marketed psychiatric drug is efficacious in approximately half of the patients who take it. One reason for this low response rate is the artificial grouping of heterogeneous syndromes with different pathophysiological mechanisms into one disorder.”
- 7. 7 What may come next: Research Domain Criteria (RDoC) • “Develop, for research purposes, new ways of classifying mental disorders based on dimensions of observable behavior and neurobiological measures.” research purposs.” • Identify fundamental components that may span multiple disorders (e.g., executive function, affect regulation) • Determine the full range of variation, from normal to abnormal • Integrate genetic, neurobiological, behavioral, environmental, and experiential components • Develop reliable and valid measures of these fundamental components for use in basic and clinical studies
- 8. 8 Exactly what does RDoC involve? • A focused research grant funding initiative (~ 40 funded to date; 14 child, 11 adult of the RFAs) • Research “toward a new classification system”: study and validate dimensional constructs that cut across current disorders • A *framework* for research – NOT a finished system • Goal: more homogeneous groupings for interventions – developing new treatments, or matching current treatments to precision assessment
- 9. The RDoC Framework: Four dimensions
- 10. • Elevated Stress Reactivity • Problems with emotion regulation [Symptoms] • Lack of pleasure in usual activities • Lack of energy for productive tasks • Memory problems • Problems with executive function • Social withdrawal • Poor relationships • Problems with arousal-modulating systems • Sleep problems
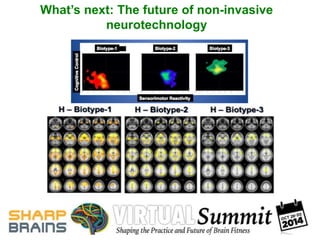
- 11. Example: BSNIP (Sz-bipolar spectrum) 0=“textbook” bipolar, 9=“txtbook Sz” Sweeney et al., SOBP Symposium, 2012 Composite Cognitive Functioning
- 12. BSNIP: Sz-bipolar spectrum (DSM analysis) Sweeney et al., 2012 NB: A significant DSM effect does not indicate meaningful differences at the individual level! Composite Cognitive Functioning
- 13. BSNIP: Sz-bipolar spectrum (RDoC approach, look at factors in better or worse cognitive function) Sweeney et al., SOBP Symposium, 2012 Composite Cognitive Functioning
- 14. BSNIP “Biotypes”: Better ways to diagnose (& treat)? Clementz, Keshavan, Pearlson, Sweeney, & Tamminga, ICOSR, 2013, shared by permisson
- 15. 15 What you should know: current approaches to validating new treatments • NIMH new Clinical Trials Program • Consistent with new trials across all of medicine • Therapeutics: major form of validation • Measurement issues, very important (for multiple units of analysis) – in a dimensional approach
- 16. Test the hypothesis about the drug’s mechanism of action: 1. Show that the treatment reaches the target, establish optimal dose (receptor occupancy or covert measures = “target engagement”) 1. Show that the treatment causes a change in relevant brain activity or mental process in the hypothesized direction (mechanism of action) 2. Correlate change in mechanism with change in clinical signal (proof of concept) 16 High Priority: Experimental Medicine, Fast-Fail Trials
- 17. What could you start doing? RDoC considerations for early phase clinical trials: 1. Focus on a novel mechanism relevant to a clinical problem 17 regardless of DSM diagnosis (e.g., anhedonia, working memory) 2. Enroll patients based on deficits in the mechanism, not DSM diagnosis 3. Both the experimental medicine paradigm and RDoC require trial outcomes that reflect the target mechanism
- 18. 18 The Latest RDoC developments • RDoC Unit in NIMH Office of the Director • RDoC as an informatics platform • Based on National Database for Autism Research (NDAR) • http://rdocdb.nimh.nih.gov • Work in progress: • (1) focused number of common data elements • (2) online discussion forum • (3) online availability of tasks and paradigms
- 19. 19 Conclusions: RDoC approach for innovation in applied neurotechnology • Standard functions (constructs) for developing new treatments • Framework for developing new constructs (or revising current ones) • Criteria for new constructs: (1) data for functional dimension of behavior/cognition; (2) data for neural circuit/system that implements the behavior • Prevents making up new targets, “gaming” the trials with measures of convenience • Can bring standardization and credibility to field • http://www.nimh.nih.gov/research-priorities/rdoc/index.shtml
- 20. What’s next: The future of non-invasive neurotechnology Charles Fisher, President of Fisher Wallace Laboratories
- 21. THE AGE OF DIGITAL PHARMA: CRANIAL ELECTROTHERAPY STIMULATION (“CES”)
- 22. Today’s Presentation • Brief History of CES • The Current Technology • Peer Reviewed Research • Clinical Applications • Q & A
- 23. Humble Beginnings of CES • 46 AD – Scribonius Largus - Black Torpedo • 1700’s – Charles Kite - electric defibrillator - reviving dead “work of the Devil” • 1850’s – Strange Contraption
- 24. Early Devices * Electro-medical apparatus, A. Gaiffe, Paris 1868
- 25. Modern CES = Digital Pharma Example: The Fisher Wallace Stimulator® • FDA Cleared to treat depression, anxiety and insomnia • Portable & Easy-to-Use • No Serious Side Effects • Use at home for 20 - 40 minutes per day • Gentle Alternating Current 1-4 mA • Frequencies: 15 hZ 500 hZ 15,000 hZ • Refined by Bernard and Saul Liss – M.I.T. / NASA
- 26. Mechanism of Action • Increases Serotonin* • Increases Beta-Endorphins* • Increases GABA* • Decreases Cortisol* • Decreases in Neuronal Activity Unlike tDCS and repetitive TMS, the effects of cranial AC stimulation seems to decrease neuronal activity, helping explain reduction in anxiety. * Proven in blood, cerebral spinal fluid & saliva tests published in peer reviewed journals
- 27. Ease of Use • Clinical Recommendations (Brown) • 20 Minutes per Treatment Session – Automatic Timer – 2X per day for Severe Symptoms (1-2 mA) • 30-45 Day Treatment Regimen • Outpatient & Inpatient • Low Maintenance – Sponges & Batteries
- 28. Patient Populations • Patients / Doctors seeking alternatives to medication • Patients with Generalized Anxiety Disorder, Major Depressive Disorder and Bipolar Disorder • Military veterans suffering from PTSD and TBI related depression, anxiety and insomnia • Women who are pregnant or nursing and therefore cannot use antidepressants or other medications • Patients who cannot tolerate the side effects of medication or are resistant to medication • Patients and doctors seeking to enhance the effect of medication (many psychiatrists prescribe our device in conjunction with antidepressant medication in order to enhance the effect of medication or to reduce medication dosage) • Patients seeking alternatives to TMS or ECT therapy • Patients suffering from mild depression, seasonal depression, jet lag, temporary anxiety, or other conditions that are not appropriately treated with long term medication • Drug and alcohol rehabilitation clinics (Phoenix House, Day Top Village, Samaritan Village)
- 29. Advantages of CES • Use With or Without Medication • Extremely Low Risk • Portable • Affordable • Easy to Use • 100% Refund Policy
- 30. PTS Symptoms are Drug Resistant A recent study published in The Journal of the American Medical Association and reported by The New Times: Antidepressants such as Risperdal, Seroquel, Geodon and Abilify that are widely prescribed to treat severe post-traumatic stress symptoms for veterans are no more effective than placebos and come with serious side effects, including weight gain and fatigue. A LOW COST, NON-DRUG THERAPY TO TREAT PTS SYMPTOMS, ESPECIALLY DEPRESSION, IS ESSENTIAL
- 31. Addiction Recovery Pilot Program • Largest Non-Profit Drug Rehab Center in USA • 392 Subjects – Cocaine and Heroine Addicts - 293 Non-CES (control) - 99 CES (for first 10 days of detox) • PTSD & Detox Symptoms: Depression, Anxiety, Insomnia
- 32. Phoenix House – Outcome • 50% increase in retention after 90 days (versus control group) • Reported profound reduction of PTSD & Detox symptoms (Anxiety & Insomnia)
- 34. New Bipolar II Study at Beth Israel Medical Center A double-blind randomized placebo controlled study of cranial electrotherapy stimulation for the treatment of depression in bipolar II disorder S Greenman BA, D McClure BA, G Kazariants PhD, M Varvara MD, S Koppolu MBBS, Z Yaseen MD, I Galynker MD PhD Beth Israel Mount Sinai Medical Center, New York, NY, USA Abstract Introduction: Cranial Electrical Stimulation (CES) technology has been used widely for treatment of depression, anxiety and insomnia, but to date, there have been no studies examining the efficacy of this technology to treat bipolar II depression. Our goal in this study was to evaluate the use of CES for treatment of the symptoms of depression in bipolar II disorder. We examined changes in levels of depression and quality of life during the four week treatment period. Methods: Patients diagnosed with bipolar II disorder and currently depressed by SCID-P were recruited from the Family Center for Bipolar in NYC. Participants were randomly assigned to either a placebo group or an active group for the first two weeks of daily 20 minute CES treatment sessions. Both groups received open-label active treatment for an additional two weeks following the randomization period. Participants were assessed at baseline, and weekly during the treatment period, using the Hamilton Depression Rating Scale (HAM-D), the Beck Depression Inventory (BDI), Clinical Global Impressions Scale (CGI-S), and the Quality of Life Satisfaction and Enjoyment Questionnaire (Q-LES-Q). ANOVAs were run to compare the groups at baseline, 1st assessment and 2nd assessment; independent t-tests to analyze differences at each time period between groups, and paired t-tests to analyze the quantitative changes between each time point. Results: The sixteen participants were 50% female, with a mean age of 47.69 (15.88), and an average level of education of 16.81 (2.401) years. Results showed a significant interaction between BDI scores and the treatment group (p =.006). There is a significant main effect of treatment group on HAM-D scores (p=.00). Groups did not differ significantly on any of the assessment measures at baseline. At week two, the active group compared to placebo had significantly higher scores on Q-LES-Q scale (p=.010) There was a significant decrease in participants’ HAM-D and BDI scores in the active group from baseline to the second week (p=.004, p=.004) and baseline to the fourth week (p=.002, p=.015).The placebo group showed a significant decrease on BDI scale scores from baseline to week four (p=.012), but no significant change from baseline to week two. In the placebo group, there was a significant change from baseline to 2 weeks on HAM-D scores, (p=.015), and during the open label treatment phase from 2 weeks to 4 weeks, (p=.022). In the active group, there was a significant decrease in severity scores using CGI-S from baseline to second week, and baseline to 4th week, (p=.017), while there is no significant difference in means for the placebo group. Discussion: Our preliminary results indicate that the active group had significantly higher reduction in depression levels compared to the placebo group. During the double-blind randomized and controlled trial of the first 2 weeks, there was a significant decrease in BDI scores for only the active group. However, the data displays improvement effects for both groups on the Hamilton Depression Rating Scale, which may be due to the difference in brain regions associated with HAM-D and BDI. Also, patients who received active treatment had decreased severity of the illness on CGI-S measures, while the placebo group had no change on the CGI-S scale. Clinical implications of this study include validating the safety and efficacy for CES in treatment of depression in bipolar II disorder. Keywords: bipolar II depression, cranial electrical stimulation This research was funded by the Fisher Wallace Laboratories. Introduction Bipolar II disorder is challenging to treat, with only marginally effective treatments. CES has been used for pain, anxiety, insomnia and depression (Kirsch & Nichols, 2013). Lack of research with controlled, randomized and double-blind trials, specifically with bipolar II patients currently depressed. Another study examining CES on bipolar disorder looked at chart reviews, N=7, and yielded no significant results. (Mostafa et al, 2013) Clinical implications include validating the safety and efficacy of Cranial Electrotherapy Stimulation for the treatment of depression. Hypothesis In this study, we hypothesize that CES will reduce depression symptom severity in the active group more than in placebo group. We also hypothesize that CES administered for 20 minutes daily for four weeks is safe and well tolerated when treating bipolar II patients. Methods Table 1. Demographic and Clinical Characteristics Safety of CES: A repeated analysis of variance was conducted on AMI, MMS, and CFQ, and no significant trends or effects were displayed. There were no changes within groups or between groups throughout the treatment. Blood Pressure: Text Mean aggregates for each week, from weeks 1-4 for active and placebo were conducted. No significant changes in systolic or diastolic pressure were found. Table 2. Side effects associated with the use of CES 16 patients diagnosed with Bipolar II Disorder and currently depressed by SCID-P & HAMD were recruited by the Family Center for Bipolar in New York City and randomly assigned to active or placebo group for the first 2 weeks of tx. Assessments of depression: HAM-D, BDI, CGI-S, Q-LESQ Cognition: AMI, MMS, CFQ Safety: pre and post treatment blood pressure, EEGS, EKGS, side effects Side effects were calculated by counting the incidences during active treatment (total=210) versus during non-active treatment (n=90). The number of participants who reported event were counted. A symptom was noted as having a change greater than or equal to one difference from pre treatment to post treatment. Side effects were rated from 1=none to 4=severe. ANOVAs were run to compare trends during phase I, and independent t- tests were used to analyze the quant itat ive differences at each t ime point . Paired t- tests were used to analyze the within group changes f rom baseline to week 1, and baseline to end of Phase I. Discussion Efficacy: Results 22.00 20.67 18.75 18.14 15.50 12.25 35.00 31.17 28.25 21.50 14.75 Results of the paired sample t test revealed that the active group experienced greater reduction in depressive symptoms, such as BDI, CGI-S, and Q-LES-Q. Both groups improved on HAM-D, possible explanation is the placebo effect or different brain regions being associated with HAM-D and BDI. Safety & Tolerability: Results support our hypothesis that CES is a safe and non-invasive course of treatment. The results indicate that there was no significant difference between groups on AMI (autobiographical memory), MMS, or CFQ during phase I. Blood pressure showed no significant changes after analysis of mean aggregates per week for four weeks. This supports our hypothesis that physical functioning and limitations due to physical functioning are not negatively impacted from use of cranial electrotherapy stimulation. Implications of this study include improving treatment for depression in bipolar II disorder. Limitations of this study include the small sample size, and the specificity of the population in regards to low external validity. Future studies replicating the safety and efficacy of CES on a larger sample size would help validate our results. Results (continued) HAM-D BDI 18.67 * 14.67 12.00 Table 3.ANOVA & T- test results * Study Design Randomization 2 weeks Sham Active Non-Responders (Open label treatment) 2 weeks Follow up Phase I Phase II Phase III Responders Responders 9.00 placebo, n=9 active, n=7 15.11 10.86 baseline First Week Second Week Figure 1.A: HAM-D scores during double-blind randomized control phase. 8.00 25.00 17.00 27.75 19.38 placebo, n=7 active, n=7 baseline First Week Second Week 5.0 4.5 4.57 4.0 3.5 55 45 35 34.33 29.22 25 15 CGI-S 4.00 4.22 Q-LESQ 48.50 placebo, n=9 active, n=7 3.71 4.33 53.50 39.33 29.44 placebo, n=9 active, n=7 4.33 baseline First Week Second Week Figure 1.B: BDI scores during double-blind randomized control phase. Figure 1.C: Q-LESQ scores during double-blind randomized control phase. P<.05 At week 2 3.0 baseline First Week Second Week Figure 1.D CGI-S scores during double-blind randomized control phase. p approaches significance, p=.067 at week 2. Characteristic Active Tx, N=7 Mean (SD) Placebo, N=9 Mean (SD) p Age 51.33(11.99) 43.78(18.26) .279 Years of Education 15.83(1.84) 17.11(2.57) .590 N(%) N(%) Employed or student 2(28.6) 2(22.22) Single/Separated/Divorced 3(42.9) 7(77.8) .329 Sex: Male 5(71.4) 3(33.3) .315 Comorbid personality disorders 6(85.7) 7(77.8) 1.00 Borderline 1(14.3) 4(44.4) .308 Narcissistic 3(42.9) 0 .063 OCPD 2(28.6) 3(33.3) 1.00 Side Effect Percent of Pat ients Report ing Event Percent of Incidences per Total Number of Treatments CES (n= 18) Placebo (n= 9) CES (n= 210) Placebo (n= 90) Drowsiness 61 66.7 8.1 10 Blurred Vision 22 22.2 4.8 7.8 Dizziness 16.7 16.7 1.9 1.1 Headache 44.4 77.8 9.5 10 References Kirsch D., Nichols F., (2013). Cranial Electrotherapy Stimulation for Treatment of Anxiety, Depresion, and Insomnia. The Psychiatry Clinics of North America. Time and treatm ent interac tion Active treatment vs. placebo Mostafa A., El-Wasify M., Elmaadawi A., Jeannie R., Mallakh R., Cranial Electrotherapy Stimulation for the Treatment of Chronically Symptomatic Bipolar Patients. (2013) Journal of ECT. Treatment effect over time Baseline Week 1 Week 2 Baseline - Week 1 Baseline - Week 2 Scales Tota l N F p Mean (SD) Mean diff. t p Mean (SD) Mean diff. t p Mean (SD) Mean diff. t p 95% CI p 95% CI p Primary Outcomes: BDI Placebo 9 27.75 (12.86) 3.42 0.52 0.62 19.38 (8.82) -1.02 -0.26 0.802 25.00 (12.07) -8.32 -2.44 0.172 [-1.01 - 17.76] 7.30E-02[-2.34 - 7.84] 0.242 Active 7 31.17 (11.43) 18.67 (6.68) 17.00 (12.28) [1.07 - 23.93] 3.80E-02 [7.01 - 21.33] 4.00E-03 time x tx group interaction 10.89 6.00E-03 HAM-D Placebo 9 18.14 (4.74) -2.52 -1 0.34 14.67 (4.47) -2.67 -1.29 0.218 15.11 (3.86) -4.25 1.7 0.112 [0.89 - 11.11] 2.70E-02[1.41 - 9.70] 1.50E-02 Active 7 20.67 (5.22) 12 (3.56) 10.86 (6.18) [3.61 - 8.67] 1.00E-03 [3.41 - 11.16] 4.00E-03 time x tx group interaction 0.489 0.496 Secondary Outcomes: CGI-S Placebo 9 4.33 (0.5) 0.24 0.92 0.38 4.22 (0.441) -0.22 -0.88 0.396 4.33 (0.5) -0.62 -1.97 6.90E-02 [-0.49 - 0.71] 0.681 [-0.54-0.54] 1 Active 7 4.57 (0.54) 4.00 (0.577) 3.71 (0.756) [0.08 - 1.07] 0.03 [0.22 - 1.5] 1.70E-02 time*tx group 5.906 2.90E-02 Q-LES Q Placebo 9 29.22 (14.84) 5.11 0.6 0.56 39.33 (18.41) 8.67 1.08 0.299 29.44 (16.13) 23.56 2.984 0.01 [-21.94 - 1.72] 8.40E-02 [-12.2 - 11.75] 0.967 Active 6 34.33 (18.21) 48.5 (12.97) 53.5 (16.40) [-24.54 - (-3.79)] 1.70E-02 [-37.8 - (-. 53)] 4.60E-02 time*tx group 4.774 4.80E-02 GAF Placebo 9 50.11 (5.93) 5.03 1.54 0.15 55.56 (7.49) -0.13 0.897 52.33 (7.68) 6.24 1.703 0.111 [-13.2 - 2.31] 0.144 [-9.55 - 5.11] 0.504 Active 7 55.14 (7.20) 55.14 (3.89) 58.57 (6.68) [-7.06 - 7.06] 1 [-7.63 - 0.77] 9.30E-02 time*tx group 9.40E-02 0.763 YMRS Placebo 9 0.78 (1.20) -0.06 -0.11 0.911 1.11 (2.03) 0.46 0.43 0.672 1.11 (1.05) 0.75 0.902 0.383 [-2.29 - 1.63] 0.705 [-1.1 - 0.44] 0.347 Active 7 0.71 (0.951) 1.57 (2.23) 1.86 (2.19) [-2.31 - 0.6] 0.2 [-2.69 - 0.41] 0.121 Friday, May 9, 14
- 35. Hamilton Depression Rating Scale
- 37. Challenges ahead for field at large • FDA regulation • Reimbursement • Distribution channels: B2B vs B2C • Integration of mature and new technologies • Proper adoption to maximize value yet ensure safety
- 38. Additional Information • www.FisherWallace.com • Chip Fisher – President • chip@fisherwallace.com • Cell: 917-912-0629
- 39. To learn more, visit SharpBrains.com
