Management of parkinson’s disease
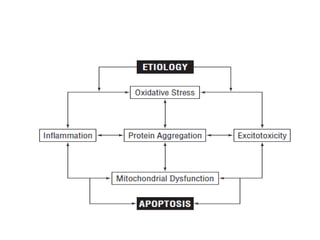
- 2. • Dopaminergic neuron loss • extensive non dopaminergic pathology - cholinergic neurons of the nucleus basalis of Meynert. - norepinephrine neurons of the locus coeruleus - serotonin neurons in the midline raphe - neurons in the cerebral cortex, brainstem, spinal cord, and peripheral autonomic nervous system.
- 4. Medications available for PD • Dopamine replacement therapy is the major medical approach. • Leading unmet need is to stop or slow progression. • Development of symptoms unresponsive to dopaminergic therapy
- 6. levodopa
- 9. L-dopa • Peripheral decarboxylase inhibitors carbidopa and benserazide. • Allows fourfold reduction in L-dopa dosage. • Immediate release, controlled release(2/3rd ) • Absorption only in small intestine. • H-pylori eradication • Malignant melanoma-intermediary in melanin synthesis • Elevated plasma homocysteine (methyl group in COMT)
- 12. Dopamine agonists • Second most powerful • Lisuride- water soluble, sc. • Cabergoline-long acting, once a day. • Rotigotine- transdermal patch • Apomorphine-sc, intranasal • Nausea, orthostatic hypotension, edema, sleep attacks,valvulopathy
- 14. COMT inhibitors • Extend half life of levodopa without increasing peak plasma concentration. • Elevated LFT, diarrohea
- 15. COMT inhibitors
- 17. MAO inhibitors
- 18. MAO -inhibitors • Cheese effect with MAO A inhibitors • Not taken with levodopa • MAO-B safe • Dual inhibitors-theoretically good
- 19. anticholinergics
- 20. amantadine
- 21. amantadine • Dopamine release • Antiglutamate • Quick onset (2 days) • Livedo retcularis, edema,visual hallucinations
- 23. Therapeutic Principles • Keep the patient functioning independently as long as possible • Patients should be encouraged to remain active and mobile • Individualize therapy • If therapies are established protective, they should be given priority
- 24. Treatment of early stage(mild symptoms,no restriction of activities • Excellent candidates for clinical trials. • Symptomatic treatment can be delayed • Neuroprotection—neuro protection neuro restoration neuro regeneration
- 25. Selegiline and antioxidants • DATATOP-selegiline and tocopherol • Selegiline delayed symptomatic treatment by 9 months • Reduced rate of worsening of UPDRS • Decreased risk of freezing • Symptomatic effect vs neuroprotective • Total MAO blockade • Tocopherol -ineffective
- 26. • Riluzole Glutamate excitotoxicity Riluzole blocks Vd sodium channel Not effective • Trophic factors GDNF-survival of neurons, dendritic growth , quantal size immunophilins- ligands primate studies encouraging
- 27. • Enhancing mitochondria and energy function-co Q 10 and creatine, not to be futile • Counteracting inflammation-minocycline, not futile • Inhibiting apoptosis-rasagiline • Dopamine agonists-CALM PD
- 28. neuroprotection
- 30. Treatment of mild stage (interferes with activities) • Symptomatic treatment. • Degree of disability and age • Severe disease-levodopa, rapid guaranteed action • Younger patients(<60 yrs),(<70 if mentally strong)- dopa sparing strategy (DA agonists, amantadine, anticholinergics) prone to develop motor complications
- 32. Dopamine agonists • Less likely to induce motor complications • Monotherapy successful for more than 3 years only in 30%. • Ergots-st anthonys fire, retroperitoneal, pleuro-pericardial fibrosis, valvulopathy • Non ergots-drowsiness, sleep attacks. • Nausea, hypotension, leg edema, hallucinations,sedation.
- 33. Dopamine agonists • Start with a tiny dose, bed time dose. • Bromocriptine 1.25,pergolide-0.05,pramipexole- 0.125 for first 3 days, ropinirole 0.25 tid for 1 st week. • Gradual dose escalation at bromocrptine-1.25 /wk, pergolide-0.125/wk,ropinirole-0.5 /day twice weekly, pramipexole 0.125/2 days for 10 days, then 0.125/day weekly. • Switching among agonists • 1:1:5:10 =pergolide:pramipexole:ropinirole:bromocriptine
- 34. amantadine • Effective in 2/3 of mildly affected patients ,rapid onset in 2 days • 100 md bid t1/2 28 hrs, max 400mg. • Dose reduction in renally impaired • Short lived effect in advanced stages but adjunctive to levodopa and agonists . • Reduces l-dopa induced dyskinesias.
- 35. anticholinergics • Monotherapy or adjunctive to l dopa/DA in tremor. • Start with low doses, trihexyphenydyl 1mg bd, benztropine 0.5mg bd • Side effects-peripheral and central • >70 yrs, weaker anticholinergic- diphenhydramine-50mg tid,orphenadrine-50mg tid,,cyclobenzaprine-20mg tid,amitriptyline 25 md tid. • Advance stages ?
- 37. Moderate stage-inadequate response to non levodopa medications • Rule of thumb-utilize the lowest drug that brings about symptom reversal. • 10/100,25/100,50/200. • Atleast 50-75 mg of cabidopa is required for adequate inhibition of peripheral decarboxylase. • If L-dopa , 300 mg/day use 25/100 form. • Carbidopa tablets are available for vomiting
- 38. Immediate release vs extended release • Rapid response with IR. • Longer half life and lower peak plasma levels with CR, unpredicatable response. • 2/3-3/4 th of identical dose of IR. • Useful in older patients, less chance of drowsiness. • Pre bed time dose allows mobility in night. • Mean of 9.3 +_1.8 days to achieve max response.
- 39. • Most common plateau schedule neurologist aim for is 25/100 mg tid. • Start with low dose 25/100,increase in weekly strengths of 25/100. • Bradykinesia and rigidity respond well than tremor. • Increase to 50/200 mg tid before adding agonist. • Before concluding ineffective -2000mg. • As disease progresses, duration of effect decreases. • Avoid sudden withdrawal
- 40. Advanced stage-motor complications,freezing,falls • All patients should be receiving levodopa therapy or atleast have had a trial of the drug but have not been able to remain on it because of disabling side effects • Fluctuations and dyskinesias • Temporal relation to last dose, sensory, motor or behavioural phenomenon
- 41. Fluctuations(offs) • Slow wearing off • Sudden off • Random off • Yo-yoing • Delayed on • Dose failure • Weak response at end of the day • Variation with meals • Sudden transient freezing Dyskinesias • Peak dose chorea,ballism,dystonia • Diphasic chorea and dystonia • Off dystonia • Myoclonus • Simultaneous dyskinesia and parkinsonism
- 42. Sensory and behavioural offs • Pain • Akathisia • Depression • Anxiety • Dysphoria • panic
- 44. pathogenesis • Advance stage of disease,duration and dose of levodopa treatment • Pharmocokinetic and pharmacodynamic mechanisms • Altered dopamine storing capacity • Dopamine receptor super sensitivity and differential sensitivity • Intermittent levodopa administration
- 46. Wearing off • Adequate dose of levodopa does not last at least 4 hrs • As plasma levels fall, clinical response also falls • Selegiline, rasagiline-decrease off time by 1.5 hrs • Comt inhibitor-increase on time by 1 hr • CR preparation-starting at night dose and working forward • Standard preparation closer intervals • Dopamine agonists • Zonisamide and adenosine antagonists.
- 47. On and off/random off • Unrelated to timing of last dose . • Careful plotting of these off periods. • Disolved levodopa in carbonated water. • Subcutaneous/intranasal apomorphine • 10-15 minutes
- 48. Dose failures/ no on • Episodic failure of patient to respond to each dose. • Poor gastric emptying. • Dissolved levodopa/apomorphine • prokinetics
- 49. Delayed on • Problem in getting an on with first dose in the morning. • Plasma levels vs low affinity • Higher morning dose to kick on • Dissolved levodopa
- 50. Weak response at end of the day • Diurnal variation • Decreased plasma levels with each dose. • Increasing afternoon or evening dose • CR preparation • DA, amantadine ,anticholinergics
- 51. Response variations with meals • Full meal-delayed gastric emptying, delayed and weaker response • Empty stomach-rapid response with dyskinesias. • High protein meals-amino acids interferes with mucosal transport of levodopa. Non protein meals in morning. Normal protein meals in night
- 52. freezing • On freezing or off freezing • Start hesitation, target hesitation, turning hesitation, startle hesitation. • Selegiline. • L-threo-dops- precursor of nor epinephrine. • Botulinum into legs of freezers
- 53. Peak dose dyskinesias • Severity of disease is the major risk factor. • Chorea is more common ,dystonia is more disabling. • Overdosed state. • Small frequent dose or CR preparation • DA with reduction of l dopa • Amantadine • Clozapine, fluoxetine, propranolol, buspirone, mirtazapine
- 54. Diphasic dyskinesias • D-I-D phenomenon • Plasma levels are falling or rising, but not during peak plasma level. • Predominantly the legs • Differential sensitivity of receptors • DA with long duration of action with small levodopa
- 55. Off dytonia • Early morning dystonia • Pharmacokinetic problem • Low plasma levels • CR preparation • Pergolide • Dystonia can also be a feature of PD, high dose or low dose
- 56. Yo-yo ing • Combination of fluctuations and dyskinesias • Plain levodopa without carbidopa • DA • Liquefied sinemet-4 tabs of 25/250 in 1 litre of soda-conc 1mg/ml,stored in dark • Intraduodenal pump
- 57. others • Myoclonus-ominous sign, DLB, levodopa toxicity,methysergide. • Somatotopically specific dyskinesias-head and neck more sensitive than legs • Loss of efficacy over time • Falling due to loss of postural reflexes- keep the patient sufficiently parkinsonian • Surgery-mouth dissolving l- dopa, apomorphine
- 58. time drug meal sleep tremor stifness others 00-1 am 1-2am 2-3am 3-4am 4-5am 5-6am 6-7am 7-8pm 8-9am 9-10am 10-11am 11-12am 12-1pm 1-2pm 2-3pm 3-4pm 4-5pm 5-6pm 6-7pm 7-8pm 8-9pm 9-10pm 10-11pm 11-00pm
- 66. dementia
- 67. psychosis
- 75. insomnia
- 76. somnolence
- 77. Future advances
- 80. Thank you
- 81. • The Scientific and clinical basis for treatment of parkinson’s disease-2009 AAN • Mark stacy-Medical treatment of PD • Stanley Fahn-Medical treatment of PD