Bond angles question answer
•Transferir como PPT, PDF•
3 gostaram•1,225 visualizações
Denunciar
Compartilhar
Denunciar
Compartilhar
Recomendados
Mais conteúdo relacionado
Mais procurados (20)
Atomic structure - Electron Configurations and Periodic Table

Atomic structure - Electron Configurations and Periodic Table
Mais de matcol
Mais de matcol (19)
Último
Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"

Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"National Information Standards Organization (NISO)
Último (20)
Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"

Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"
JAPAN: ORGANISATION OF PMDA, PHARMACEUTICAL LAWS & REGULATIONS, TYPES OF REGI...

JAPAN: ORGANISATION OF PMDA, PHARMACEUTICAL LAWS & REGULATIONS, TYPES OF REGI...
Call Girls in Dwarka Mor Delhi Contact Us 9654467111

Call Girls in Dwarka Mor Delhi Contact Us 9654467111
Interactive Powerpoint_How to Master effective communication

Interactive Powerpoint_How to Master effective communication
Z Score,T Score, Percential Rank and Box Plot Graph

Z Score,T Score, Percential Rank and Box Plot Graph
The byproduct of sericulture in different industries.pptx

The byproduct of sericulture in different industries.pptx
A Critique of the Proposed National Education Policy Reform

A Critique of the Proposed National Education Policy Reform
Disha NEET Physics Guide for classes 11 and 12.pdf

Disha NEET Physics Guide for classes 11 and 12.pdf
Bond angles question answer
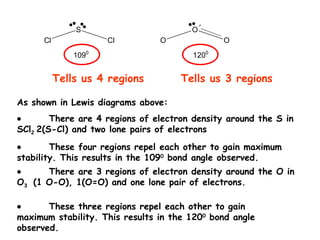
- 1. Tells us 4 regions Tells us 3 regions As shown in Lewis diagrams above: There are 4 regions of electron density around the S in SCl 2 2(S-Cl) and two lone pairs of electrons These four regions repel each other to gain maximum stability. This results in the 109 0 bond angle observed. There are 3 regions of electron density around the O in O 3 (1 O-O), 1(O=O) and one lone pair of electrons. These three regions repel each other to gain maximum stability. This results in the 120 0 bond angle observed.