Elements and Compounds 2
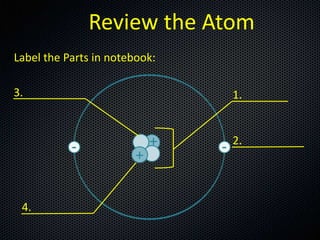
- 1. Review the Atom Label the Parts in notebook: 3. 1. 2. 4.
- 2. Review the Atom Label the Parts in notebook: 3. Neutron 1. Nucleus 2. Electron 4. Proton
- 3. Elements and Compounds 2 Foundation Science 4 Class Code SC22102
- 4. Molecules A molecule is 2 or more atoms connected to each other
- 5. Compounds A compound is 2 or more DIFFERENT atoms connected to each other
- 6. Practice Answer molecules, compounds, or both in your foundation notebook. 1. 2. 3. 4. 5. 6. 7. 8.
- 7. Answer Questions in Notebook 1. 2. 3. 4. 5. A ________ is 2 or more atoms connected to each other. A ________is 2 or more DIFFERENT atoms connected to each other _________ is anything that has mass and takes up space. An ________ is the basic unit of matter. When something is made up of only one type of atom, it is called an ___________. 6. The ________ __________ is the number of protons in an atom. 7. Water (H2O) is a ______ (compound, molecule or both) 8. Nitrogen (N2) is a _______ (compound , molecule or both)
- 8. Answers 1. 2. 3. 4. 5. A molecule is 2 or more atoms connected to each other. A compound is 2 or more DIFFERENT atoms connected to each other Matter is anything that has mass and takes up space. An atom is the basic unit of matter. When something is made up of only one type of atom, it is called an element. 6. The atomic number is the number of protons in an atom. 7. Water (H2O) is a both (compound, molecule, both) 8. Nitrogen (N2) is a molecule (compound, molecule, both)
- 9. Elements in the Human Body The human body is made up mostly of Oxygen (65%), Carbon (18%) and Hydrogen (10%). We are about 70% water (= H and O) The body also has: 3.2% Nitrogen 1.5% Calcium 1.0% Phosphorus
- 10. Elements in the Earth’s Crust Earth’s crust is the outer layer… Crust is: 50% Oxygen (O) 26% Silicon (Si) 7% Aluminum (Al) 4% Iron (Fe)
- 11. Atoms Like to Give or Share Electrons This bonds them together This creates a NEW substance called a molecule, with new properties Sodium (Na) metal is very dangerous and explodes in water Chlorine (Cl) is a very dangerous gas
- 12. Sodium (Na)
- 13. Atoms Like to Give or Share Electrons This bonds them together This creates a NEW substance called a molecule, with new properties Sodium (Na) metal is very dangerous and explodes in water + Chlorine (Cl) is a very dangerous gas But together, table salt (NaCl) is completely safe
- 14. Types of Chemical Bonds Covalent Bond – atoms share outer electrons Ionic Bond – electron moves from one atom to another
- 15. Examples of Ionic and Covalent Bonds Water (H2O) is a covalent bond Sodium chloride (NaCl) is an ionic bond
- 16. Classification of Elements Elements are classified into 3 states: Solid Silver Liquid Solid Gas Bromine Liquid Chlorine Gas
- 17. Chemical Formulas The chemical formula shows what atoms and how many are present in the compound. Example… H2O Atomic symbols How many hydrogen atoms present If there is no number it means there is 1 atom
- 18. Chemical Formulas Practice with using chemical formulas: Alcohol C2H5OH What atoms are present in this compound? Carbon (C), Hydrogen (H) and Oxygen (O) How many of each atom is present? 2 carbon atoms 6 Hydrogen atoms 1 oxygen atom
- 19. 1. 2. 3. 4. 5. 6. 7. 8. Answer In Your Notebook What is the most common element in the human body (by mass)? What is the most common element in the earth’s crust (by mass)? Is sodium metal (Na) a safe material? Is chlorine gas (Cl) safe to breathe? Is sodium chloride (NaCl) safe? A ________ bond shares its electrons. In a _________ bond electron(s) go from one atom to another. How many atoms of each element are present in the compounds: Refined sugar, C12H22O11 Sulphuric acid, H2SO4 Glucose, C6H12O6 Urea, CO (NH2)2 9. Which compound from above is this? 10. What is the chemical formula for butane?