LA Poster
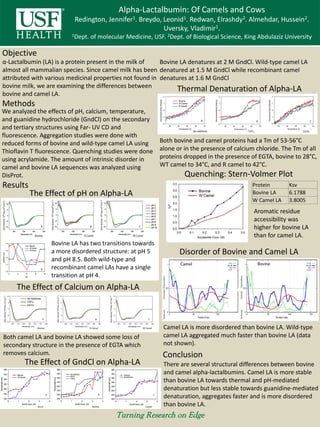
- 1. Alpha-Lactalbumin: Of Camels and Cows Redington, Jennifer1. Breydo, Leonid1. Redwan, Elrashdy2. Almehdar, Hussein2. Uversky, Vladimir1. 1Dept. of molecular Medicine, USF. 2Dept. of Biological Science, King Abdulaziz University Objective Methods Results Conclusion α-Lactalbumin (LA) is a protein present in the milk of almost all mammalian species. Since camel milk has been attributed with various medicinal properties not found in bovine milk, we are examining the differences between bovine and camel LA. We analyzed the effects of pH, calcium, temperature, and guanidine hydrochloride (GndCl) on the secondary and tertiary structures using Far- UV CD and fluorescence. Aggregation studies were done with reduced forms of bovine and wild-type camel LA using Thioflavin T fluorescence. Quenching studies were done using acrylamide. The amount of intrinsic disorder in camel and bovine LA sequences was analyzed using DisProt. There are several structural differences between bovine and camel alpha-lactalbumins. Camel LA is more stable than bovine LA towards thermal and pH-mediated denaturation but less stable towards guanidine-mediated denaturation, aggregates faster and is more disordered than bovine LA. The Effect of pH on Alpha-LA Disorder of Bovine and Camel LA The Effect of Calcium on Alpha-LA The Effect of GndCl on Alpha-LA Thermal Denaturation of Alpha-LA Protein Ksv Bovine LA 6.1788 W Camel LA 3.8005 Quenching: Stern-Volmer Plot Bovine LA has two transitions towards a more disordered structure: at pH 5 and pH 8.5. Both wild-type and recombinant camel LAs have a single transition at pH 4. Bovine LA denatures at 2 M GndCl. Wild-type camel LA denatured at 1.5 M GndCl while recombinant camel denatures at 1.6 M GndCl Both camel LA and bovine LA showed some loss of secondary structure in the presence of EGTA which removes calcium. Both bovine and camel proteins had a Tm of 53-56°C alone or in the presence of calcium chloride. The Tm of all proteins dropped in the presence of EGTA, bovine to 28°C, WT camel to 34°C, and R camel to 42°C. Aromatic residue accessibility was higher for bovine LA than for camel LA. Camel LA is more disordered than bovine LA. Wild-type camel LA aggregated much faster than bovine LA (data not shown).