Ensuring Optimal Management Throughout the Clinical Spectrum of Spinal Muscular Atrophy
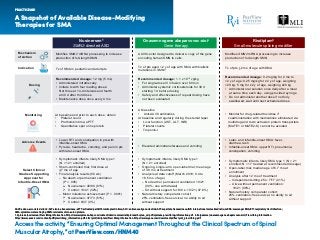
- 1. A Snapshot of Available Disease-Modifying Therapies for SMA PRACTICE AID Access the activity, “Ensuring Optimal Management Throughout the Clinical Spectrum of Spinal Muscular Atrophy,” at PeerView.com/HNM40 AAV9: adeno-associated virus 9; ALT: alanine transaminase; aPTT: activated partial thromboplastin time; ASO: antisense oligonucleotide; AST: aspartate transaminase; INR: international normalized ratio; mRNA: messenger RNA; RTI: respiratory tract infection; SMA: spinal muscular atrophy; SMN: survival motor neuron protein. 1. Spinraza (nusinersen) Prescribing Information. https://www.spinraza-hcp.com/content/dam/commercial/spinraza/hcp/en_us/pdf/spinraza-prescribing-information.pdf. 2. Zolgensma (onasemnogene abeparvovec-xioi) Prescribing Information. https://www.avexis.com/content/pdf/prescribing_information.pdf. 3. Evrysdi (risdiplam) Prescribing Information. https://www.gene.com/download/pdf/evrysdi_prescribing.pdf. Nusinersen1 SMN2-directed ASO Onasemnogene abeparvovec-xioi2 Gene therapy Mechanism of Action Indication Dosing Select Clinical Studies Supporting Approval for Infantile-Onset SMA Adverse Events Monitoring Modifies SMN2 mRNA processing to increase production of full-length SMN Tx of SMA in pediatric and adult pts Recommended dosage: 12 mg (5 mL) • Administered intrathecally • Initiate tx with four loading doses: first three at 14-d intervals and fourth at 30 d after third dose • Maintenance dose once every 4 mo At baseline and prior to each dose, obtain • Platelet count • Prothombin time; aPTT • Quantitative spot urine protein • Symptomatic infants, likely SMA type 1 (N = 121 enrolled) • Sham-controlled trial; first dose at 7 mo of age • Final analysis results (56 wk) – No death or permanent ventilation (P = .005) Nusinersen: 49/80 (61%) Control: 13/41 (32%) – Motor milestone achievement (P < .0001) Nusinersen: 37/73 (51%) Control: 0/37 (0%) • Lower RTI and constipation in pts with infantile-onset SMA • Pyrexia, headache, vomiting, and pain in pts with later-onset SMA AAV9 vector designed to deliver a copy of the gene encoding human SMN to cells Tx of pts aged <2 y of age with SMA with biallelic mutations in SMN1 Recommended dosage: 1.1 x 1014 vg/kg • For single-dose IV infusion over 60 min • Administer systemic corticosteroids for 30 d starting 1 d before dosing • Safety and effectiveness of repeat dosing have not been evaluated At baseline • Anti-AAV9 antibodies At baseline and regularly during the steroid taper Liver function (AST, ALT, INR) Platelet counts • Symptomatic infants, likely SMA type 1 (N = 21 enrolled) • Ongoing, single-arm, open-label trial; mean age of 3.9 mo at treatment • Analysis at data cutoff (March 2019; 9.4 to 18.5 mo of age) – No death or permanent ventilation: 19/21 (90%; one withdrawal) – Sit without support for 30 s: 10/21 (47.6%) • Natural history comparator cohort: 25% ventilation-free survival, no ability to sit without support • Elevated aminotransferases and vomiting Risdiplam3 Small-molecule splicing modifier Modifies SMN2 mRNA processing to increase production of full-length SMN Tx of pts 2 mo of age with SMA Recommended dosage: 0.2 mg/kg for 2 mo to <2 y of age; 0.25 mg/kg for ≥2 y of age, weighing <20 kg; 5 mg for ≥2 y of age, weighing ≥20 kg • Administer oral solution once daily after a meal at same time each day, using provided syringe • Do not administer another dose if not fully swallowed; wait until next scheduled dose • Monitor for drug-related toxicities if coadministration with medications eliminated via multidrug and toxin extrusion protein transporters (MATE1 or MATE2-K) cannot be avoided • Symptomatic infants, likely SMA type 1 (N = 21 enrolled; N = 17 treated at recommended dosage) • Open-label trial; median age of 6.7 mo at enrollment • Analysis after 12 mo of treatment – Independent sitting 5 s: 7/17 (41%) – Alive without permanent ventilation: 19/21 (90%) • Natural history comparator cohort: 25% ventilation-free survival, no ability to sit without support • Later- and infantile-onset SMA: fever, diarrhea, rash • Infantile-onset SMA: upper RTI, pneumonia, constipation, vomiting • • Troponin-I•
- 2. Monitoring and Treatment Recommendations for SMA1,2 PRACTICE AID Access the activity, “Ensuring Optimal Management Throughout the Clinical Spectrum of Spinal Muscular Atrophy,” at PeerView.com/HNM40 Respiratory Recommendations Goals Assessments Interventions Proactive approach • Earlier introduction of treatments to support airway clearance and ventilation • Physical examination • Hypoventilation (end tidal CO2 ) • Sleep study or pneumograms (symptomatic patients, to assess need for NIV) • Gastroesophageal reflux • Airway clearance – Oral suctioning – Physiotherapy/respiratory therapy (manual chest therapy, cough insufflator/exsufflator) • Ventilation: bilevel NIV in all symptomatic patients • Medication: nebulized bronchodilators if asthma suspected • Cough function • Physical examination • Sleep study or pneumograms (symptomatic patients, to assess need for NIV) • Gastroesophageal reflux • Spirometry • Airway clearance – Physiotherapy/respiratory therapy (manual chest therapy, cough insufflator/exsufflator) • Ventilation: bilevel NIV in all symptomatic patients • Medication: nebulized bronchodilators if asthma suspected • Spirometry • Airway clearance – Physiotherapy/respiratory therapy (manual chest therapy, cough insufflator/exsufflator) • Ventilation: bilevel NIV in all symptomatic patients • Medication: nebulized bronchodilators if asthma suspected NonsittersSittersAmbulatory
- 3. Monitoring and Treatment Recommendations for SMA1,2 PRACTICE AID Access the activity, “Ensuring Optimal Management Throughout the Clinical Spectrum of Spinal Muscular Atrophy,” at PeerView.com/HNM40 Rehabilitation Recommendations Goals Assessments Interventions • Optimize function and positional tolerance • Minimize impairment • Posture, scoliosis, hip dislocation, sitting tolerance, chest deformities • Contractures • Muscle weakness, functional and motor development scales • Positioning/bracing (postural, thoracic, cervical) • Stretching (orthoses) • Promote function/mobility (mobility systems, adapted equipment/toys) • Spine deformity management: changing scenario • Prevent contractures, scoliosis • Promote function/mobility • Posture, foot and chest deformities, scoliosis, hip dislocation • Contractures • Muscle weakness, functional scales • Positioning/bracing (thoracic, cervical) • Stretching (orthoses) • Promote function/mobility (mobility systems or devices based on individual ability, exercise) • Spine deformity management: orthoses in growing children; “growth friendly” rods; rods (skeletally mature) • Promote function, mobility, joint range, balance, endurance • Mobility, timed tests, endurance, functional scales, muscle weakness, falls • Contractures • Posture, scoliosis, hip dislocation • Promote function/mobility (aerobic and general conditioning exercise monitored by therapist) • Stretching (assisted, orthoses as needed) • Positioning/bracing (lower limb, thoracic) • Scoliosis surgery NonsittersSittersAmbulatory
- 4. Monitoring and Treatment Recommendations for SMA1,2 PRACTICE AID Access the activity, “Ensuring Optimal Management Throughout the Clinical Spectrum of Spinal Muscular Atrophy,” at PeerView.com/HNM40 NIV: noninvasive ventilation; SMA: spinal muscular atrophy. 1. Mercuri E et al. Neuromuscul Disord. 2018;28:103-115. 2. Finkel RS et al. Neuromuscul Disord. 2018;28:197-207. Nutritional Recommendations Caregiver Resources Swallowing and feeding difficulties/safe swallowing Appropriate calorie intake/weight control Monitor and treat gastrointestinal dysfunction • Monitor (eg, swallow tests, choking or coughing) • Feeding tube may be necessary for total or supplemental nutrition • Avoid undernutrition • Monitor fasting times during acute care episodes • Examples: reflux, constipation, use of bowel-regulating agents, delayed gastric emptying, vomiting • Monitor (eg, swallow tests, choking or coughing) • Feeding tube may be necessary for total or supplemental nutrition • Avoid undernutrition • Avoid overweight/obesity • Monitor fasting times during acute care episodes • Examples: reflux, constipation, use of bowel-regulating agents, delayed gastric emptying, vomiting • Not usually a problem in walkers • Avoid overweight/obesity • Monitor fasting times during acute care episodes • Examples: reflux, constipation, use of bowel-regulating agents, delayed gastric emptying, vomiting NonsittersSittersAmbulatory CureSMA: https://www.curesma.org/ SMA Foundation: https://smafoundation.org/ Muscular Dystrophy Association: https://www.mda.org/ TREAT-NMD: https://treat-nmd.org/ National Organization for Rare Disorders: https://rarediseases.org/ Rare Diseases Clinical Research Network: https://www.rarediseasesnetwork.org/
- 5. Access the activity, “Ensuring Optimal Management Throughout the Clinical Spectrum of Spinal Muscular Atrophy,” at PeerView.com/HNM40 Motor Function Assessment Scales for SMA1-4 PRACTICE AID Hammersmith Infant Neurological Examination (HINE) HINE Section 2: Motor Milestonesa Simple and scorable method for evaluating infants from 2 mo to 2 y of age Section 1: Neurologic exam assessing cranial nerve function, posture, movements, tone, reflexes, reactions Section 2: Motor milestones (head control, sitting, voluntary grasp, ability to kick, rolling, crawling, standing, walking) Section 3: Behavioral assessment (state of consciousness, emotional state, social orientation) Score 0 1 2 3 4 Head control Unable to keep head upright Wobbles (4 mo) Keeps upright all the time (5 mo) Sitting Cannot sit Sits with support at hips (4 mo) Props self up (6 mo) Stable sitting (7-8 mo) Pivots (9 mo) Voluntary grasp No grasp Uses whole hand Index finger and thumb but immature grasp Pincer grasp Ability to kick No kicking Kicks horizontally but legs do not lift Upward (vertical; 3 mo) Touches leg (4-5 mo) Touches toes (5-6 mo) Rolling No rolling Rolls to side (4 mo) Prone to supine (6 mo) Supine to prone (6 mo) Crawling or bottom shuffling Does not lift head On elbow (3 mo) On outstretched hand (4 mo) Crawls flat on abdomen (8 mo) Crawls on hands/knees (10 mo) Standing Does not support weight Supports weight (4 mo) Stands with support (7 mo) Stands unaided (12 mo) Walking Bouncing (4 mo) Cruising (12 mo) Walking independently (15 mo) a Normal achievement age is in parentheses.
- 6. Access the activity, “Ensuring Optimal Management Throughout the Clinical Spectrum of Spinal Muscular Atrophy,” at PeerView.com/HNM40 Motor Function Assessment Scales for SMA1-4 PRACTICE AID Children’s Hospital of Philadelphia Infant Test of Neuromuscular Disorders (CHOP INTEND) Hammersmith Functional Motor Scale—Expanded (HFMSE) Valid for motor assessment of children ranging in age from 3.8 mo to over 4 y • Developed in the context of SMA assessment • Each item graded on a scale of 0-4, with 0 as “no response” and 4 as “complete response” • Total score ranges from 0 to 64 Spontaneous movement (upper extremity) Hip adductors Shoulder flexion and elbow flexion Elbow flexion Sitting Items 1-4 Rolling Items 5-9 Transitions/ crawling Items 10-17 Standing/ stepping Items 18-20 Transitions/ kneeling Items 21-27 Squat/ jump Items 28-29 Stairs Items 30-33 Hand grip Rolling: elicited from arms Hip flexion and foot dorsiflexion Head/neck extension Spontaneous movement (lower extremity) Rolling: elicited from legs Knee extension Neck flexion Head in midline with visual stimulation Shoulder and elbow flexion and horizontal abduction Head control Spinal incurvation Evaluates motor function in individuals with later-onset SMA • Each item scored on a scale of 0-2, with patient fatigue an important consideration • Total score ranges from 0 to 66, with lower scores indicating poorer motor function
- 7. Access the activity, “Ensuring Optimal Management Throughout the Clinical Spectrum of Spinal Muscular Atrophy,” at PeerView.com/HNM40 Motor Function Assessment Scales for SMA1-4 PRACTICE AID SMA: spinal muscular atrophy. 1. https://smauk.org.uk/physiotherapy-based-measures-hine-chop-intend-hmfse-for-people-with-sma-and-how-they-are-used-to-monitor-treatment-outcomes. 2. Mazzone E et al. Neuromuscul Disord. 2011;21:406-412. 3. Montes J et al. Neurology. 2010;74:833-838. 4. https://www.who.int/childgrowth/standards/motor_milestones/en/. World Health Organization (WHO) Motor Development Milestones Explores the windows of achievement of six gross motor milestones for children aged 4 to 24 mo Sitting without support Standing with assistance Crawling on hands and knees Walking with assistance Standing alone Walking alone 6-Minute Walk Test (6MWT) Evaluates exercise capacity in ambulatory individuals with later-onset SMA • Test course has start line, with horizontal lines placed every 1 m; running/jogging not permitted • Participants walk as fast as possible along 25-m course on flat linoleum surface, turn around marker cone, return in opposite direction, repeating loop as often as possible for 6 min 25 m Upper Limb Module (ULM) Assesses functions related to everyday life in nonambulatory individuals with SMA aged 30 mo to 27 y Pencil Coins Push light Container lid Paper Plastic cups Weights
- 8. Diagnostic Testing for SMA1-3 PRACTICE AID Access the activity, “Ensuring Optimal Management Throughout the Clinical Spectrum of Spinal Muscular Atrophy,” at PeerView.com/HNM40 NM: neuromuscular; SMA: spinal muscular atrophy; SMN: survival motor neuron. 1. Mercuri E et al. Neuromuscul Disord. 2018;28:103-115. 2. Glascock J et al. J Neuromuscul Dis. 2018;5:145-158. 3. Glascock J et al. J Neuromuscul Dis. 2020;7:97-100. • Additional tests (eg, electromyography, nerve conduction tests) are not usually required for diagnosis Clinical suspicion of SMA 0 copies Other SMA or NM disorders SMN1 Confirmatory testing (SMN1 deletion testing) 1 copy ≥2 copies Confirmed 5q SMA Mutation No mutation Predictive value of SMA severity Allows immediate inclusion in therapies SMN2 copy #1 (probable type 0): treatment based on symptoms SMN2 copy #2 (probable type 1): treat SMN2 copy #3 (probable type 2/3): treat SMN2 copy #4 (probable type 3/4): treat SMN1 sequencing Positive newborn screening SMN2
