Atoms, Elements, Compounds, Mixtures, GLS.pptx
•Transferir como PPTX, PDF•
0 gostou•151 visualizações
Atoms, Elements, Compounds, Mixtures State of matter
Denunciar
Compartilhar
Denunciar
Compartilhar
Recomendados
Team Lead Succeed – Helping you and your team achieve high-performance teamwo...

Team Lead Succeed – Helping you and your team achieve high-performance teamwo...Association for Project Management
Mattingly "AI & Prompt Design: Large Language Models"

Mattingly "AI & Prompt Design: Large Language Models"National Information Standards Organization (NISO)
Mais conteúdo relacionado
Último
Team Lead Succeed – Helping you and your team achieve high-performance teamwo...

Team Lead Succeed – Helping you and your team achieve high-performance teamwo...Association for Project Management
Mattingly "AI & Prompt Design: Large Language Models"

Mattingly "AI & Prompt Design: Large Language Models"National Information Standards Organization (NISO)
Último (20)
Using Grammatical Signals Suitable to Patterns of Idea Development

Using Grammatical Signals Suitable to Patterns of Idea Development
Decoding the Tweet _ Practical Criticism in the Age of Hashtag.pptx

Decoding the Tweet _ Practical Criticism in the Age of Hashtag.pptx
Team Lead Succeed – Helping you and your team achieve high-performance teamwo...

Team Lead Succeed – Helping you and your team achieve high-performance teamwo...
Mattingly "AI & Prompt Design: Large Language Models"

Mattingly "AI & Prompt Design: Large Language Models"
4.11.24 Mass Incarceration and the New Jim Crow.pptx

4.11.24 Mass Incarceration and the New Jim Crow.pptx
MS4 level being good citizen -imperative- (1) (1).pdf

MS4 level being good citizen -imperative- (1) (1).pdf
Mental Health Awareness - a toolkit for supporting young minds

Mental Health Awareness - a toolkit for supporting young minds
Destaque
More than Just Lines on a Map: Best Practices for U.S Bike Routes

More than Just Lines on a Map: Best Practices for U.S Bike RoutesProject for Public Spaces & National Center for Biking and Walking
Destaque (20)
Content Methodology: A Best Practices Report (Webinar)

Content Methodology: A Best Practices Report (Webinar)
How to Prepare For a Successful Job Search for 2024

How to Prepare For a Successful Job Search for 2024
Social Media Marketing Trends 2024 // The Global Indie Insights

Social Media Marketing Trends 2024 // The Global Indie Insights
Trends In Paid Search: Navigating The Digital Landscape In 2024

Trends In Paid Search: Navigating The Digital Landscape In 2024
5 Public speaking tips from TED - Visualized summary

5 Public speaking tips from TED - Visualized summary
Google's Just Not That Into You: Understanding Core Updates & Search Intent

Google's Just Not That Into You: Understanding Core Updates & Search Intent
The six step guide to practical project management

The six step guide to practical project management
Beginners Guide to TikTok for Search - Rachel Pearson - We are Tilt __ Bright...

Beginners Guide to TikTok for Search - Rachel Pearson - We are Tilt __ Bright...
Unlocking the Power of ChatGPT and AI in Testing - A Real-World Look, present...

Unlocking the Power of ChatGPT and AI in Testing - A Real-World Look, present...
More than Just Lines on a Map: Best Practices for U.S Bike Routes

More than Just Lines on a Map: Best Practices for U.S Bike Routes
Ride the Storm: Navigating Through Unstable Periods / Katerina Rudko (Belka G...

Ride the Storm: Navigating Through Unstable Periods / Katerina Rudko (Belka G...
Atoms, Elements, Compounds, Mixtures, GLS.pptx
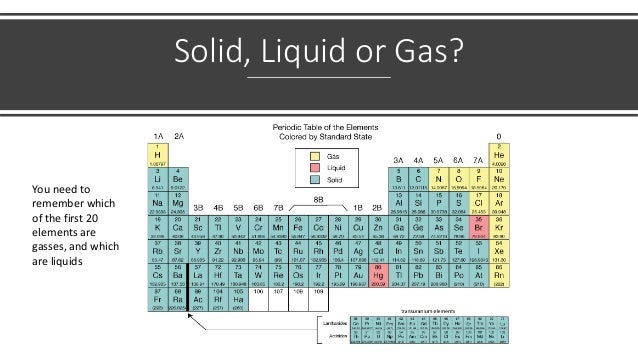
- 1. Solid, Liquid or Gas? You need to remember which of the first 20 elements are gasses, and which are liquids
- 2. Compounds and Mixtures • All substances are either elements, compounds or mixtures. • An element is made up of only one type of atom. Carbon and oxygen are both elements. • A compound is a substance made up of two or more elements joined together. For example, when carbon atoms join with 2 oxygen atoms, they form a compound called carbon dioxide. • Carbon atoms that are just mixed with oxygen atoms, but are not attached to them, form a mixture. A mixture can also contain compounds, if they are not joined to the other substances in the mixture. • For example, air is a mixture of elements such as nitrogen and oxygen, and compounds, such as water and carbon dioxide.
- 3. “Stable” atoms/elements? Atoms are more stable energetically when they have eight electrons in their valence shell, the outermost electron shell. Atoms ‘want’ a full outer shell of electrons, i.e. they are most stable with a full outer shell. This explains why the Group 8 elements, the noble gases, do not react to make compounds — they are already as stable as they can be