Amy Lane Pacifichem Biosynthesis Natural Products
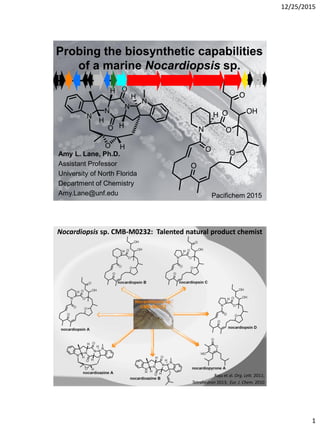
- 1. 12/25/2015 1 Amy L. Lane, Ph.D. Assistant Professor University of North Florida Department of Chemistry Amy.Lane@unf.edu Probing the biosynthetic capabilities of a marine Nocardiopsis sp. regulationortransportoxidoreductase nsnR1 R2 I H T1T2 orf7 cytochromeP450 F L hypotheticalorother E nonribosomalpeptide synthetase(NRPS) A B C D G epoxidehydrolase lysinecyclodeaminase R3 R4 orf17-18 polyketide synthase(PKS) Pacifichem 2015 Raju et al. Org. Lett. 2011; Tetrahedron 2013; Eur. J. Chem. 2010 Nocardiopsis sp. CMB-M0232: Talented natural product chemist
- 2. 12/25/2015 2 Nocardiopsins: Newest rapamycin class natural products • FKBP modulators. • Nocardiopsins structurally distinct from other class members. FK506 rapamycin nocardiopsin A Vezina et al. (1975) J. Antibiot.; Kino et al. (1987) J. Antibiot.; Raju et al. (2010) Eur. J. Chem. Nocardiopsins: Newest rapamycin class natural products FK506 rapamycin nocardiopsin A Vezina et al. (1975) J. Antibiot.; Kino et al. (1987) J. Antibiot.; Raju et al. (2010) Eur. J. Chem. Macrolide ring closure via 3o C-OH with non-PKS derived hydroxy group THF group
- 3. 12/25/2015 3 Nocardiopsin Polyketide Core Assembly Bis et al. ChemBioChem (2015) regulation or transportoxidoreductase nsnR1 R2 I H T1 T2 orf7 cytochrome P450 F L hypothetical or other E nonribosomal peptide synthetase (NRPS) A B C D G epoxide hydrolase lysine cyclodeaminase R3 R4 orf17-18 polyketide synthase (PKS) N O O O O O OH OH5 6 N O O O O O OH OH 10 Oxidoreductase and/or P450 (NsnH, NsnI) nocardiopsin B 6 8 Nonribosomal peptide incorporation and macrolide formation Bis et al. ChemBioChem (2015) regulation or transportoxidoreductase nsnR1 R2 I H T1 T2 orf7 cytochrome P450 F L hypothetical or other E nonribosomal peptide synthetase (NRPS) A B C D G epoxide hydrolase lysine cyclodeaminase R3 R4 orf17-18 polyketide synthase (PKS) NH2 COOHH2N lysine cyclodeaminase (NsnL) N H COOH L-lysine L-pipecolic acid NRPS, L-pipecolic acid NRPS (NsnE) Suggests novel biosynthetic route to prepare molecule for macrolide closure. nsnG nsnF
- 4. 12/25/2015 4 Nocardioazines: Anticancer natural products from a marine Nocardiopsis sp. • Uniquely prenylated indole alkaloid diketopiperazine (DKP). • New class of bridged DKP. • Reverse drug resistance in cancer cells by inhibiting P-glycoprotein. Raju et al. (2011) Org. Lett. Probing the enzymology of the nocardioazine biosynthetic pathway Biosynthesis of cyclo(Trp-Trp) core: Hypothesis 1: Core assembled via nonribosomal peptide synthetase. Hypothesis 2: Core assembled via cyclodipeptide synthase (CDPS). Two putative CDPSs encoded by genome. James et al. ACS Synthetic Biology. Articles ASAP.
- 5. 12/25/2015 5 Cyclodipeptide synthases (CDPSs): Candidates for Biosynthesis of Nocardioazine Core • First CDPS was characterized by Gondry and coworkers in 2009. Reviewed by Belin et al. (2012) Nat. Prod. Rep. CDPS-encoding gene clusters from Nocardiopsis sp. • The noz cluster encodes CDPS as well as additional enzymes with putative roles in nocardioazine assembly. • The ncd cluster encodes only a CDPS homolog and no other enzymes homologous to biosynthetic enzymes. James et al. ACS Synthetic Biology. Articles ASAP.
- 6. 12/25/2015 6 time (min) • Expression of either NozA or NcdA in E. coli resulted in biosynthesis of cyclo(Trp-Trp). • No other DKP products were observed. • In vitro assays with purified enzymes also supported tryptophanyl-tRNA as sole substrate of both CDPSs. Evaluating the biosynthetic function of NozA and NcdA A James et al. ACS Synthetic Biology. Articles ASAP Both nozA and ncdA are transcribed by Nocardiopsis sp. • RT-PCR suggests both NozA and NcdA catalyze production of cyclo(L-Trp-L-Trp) by Nocardiopsis sp. • NozA and NcdA provide uncommon example of multiple phylogenetically distinct enzymes from one organism yielding the same secondary metabolite. James et al. ACS Synthetic Biology. Articles ASAP. RT-PCR Accumulation of cWW in Nocardiopsis cultures
- 7. 12/25/2015 7 Conclusions • The unique ensemble of natural products of Nocardiopsis sp. CMB- M0232 is assembled by an equally unique suite of biosynthetic enzymes. • NozA and NcdA catalyze formation of cyclo(L-Trp-L-Trp) and add to the group of substrate specific CDPSs. • Both NozA and NcdA are candidates for biosynthesis of cyclo(L-Trp-L- Trp) as a nocardioazine precursor. • Nocardiopsin assembly includes an unprecedented biosynthetic strategy to prepare the linear PKS-NRPS precursor for macrolide ring closure. Acknowledgements Funding: • Research Corporation Cottrell College Science Award • UNF Transformative Learning Opportunities Grants • UNF Faculty Fellowship Award Lane lab students Collaborators • Dr. Robert Capon (U Queensland) • Dr. Rajesh Viswanathan (Case Western Reserve U) • Dr. Jonathan Karty (Indiana U) • Dr. Bryan Knuckley (UNF) Elle James Dana Bis Yang Ban