Inverse-square law, sources of radiation and shielding explained
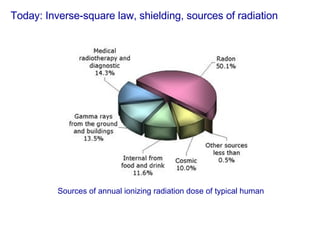
- 1. Today: Inverse-square law, shielding, sources of radiation Sources of annual ionizing radiation dose of typical human
- 7. The intensity of radiation follows the inverse square law Force Charge1 * Charge2 distance 2 Intensity of radiation Source intensity distance 2 Remember for electric charge: If you double the distance, Intensity decreases by a factor of 4
- 8. Inverse-square law demo Three pieces together are a “Geiger counter” Geiger-Müller tube High Voltage Power Supply Counter Radioactive Source Holder Calibrated distance rail
- 9. Refresher on Geiger counter Depending on the design: Maybe detect alpha-radiation CAN detect beta-radiation Maybe detect gamma-radiation Alpha particles difficulty passing through window But can make it through mica Ionizing radiation (beta, gamma, some alpha) enter through window and create ions inside. These events are detected via the electric current and turned into audible “clicks.”
- 10. Inverse-square law demo Geiger-Müller tube Radioactive Source Holder Calibrated distance rail Let’s try the experiment again from last week Distance
- 11. Three kinds of radiation emitted via radioactive decay named before they were understood…alpha, beta, gamma Alpha particles stopped by paper alpha particles are helium nuclei Beta particles stopped by aluminum beta particles are electrons (or positrons) Gamma rays stopped by lead gamma rays are high energy photons x-rays similar, but lower energy than gamma
- 12. Shielding demo Geiger-Müller tube Radioactive Source Holder Calibrated distance rail Let’s try experiments with different shielding materials Place different shielding materials here
- 27. Parallel of electrical energy with nuclear energy Electricity and radioactivity have existed since the beginning of time So, the dangers have always been present, and always will be. Humans have learned how to harness the powers of electricity and nuclear reactions…presenting new dangers along with great benefits The task for society is to weigh the benefits against the dangers Hey toddlers! I’m your friend!
- 28. How to read the short-hand summary of an isotope Number of protons Atomic number (Z) Total number of nucleons (protons + neutrons) Mass number (A) Element symbol The atomic number and element symbol provide the same information. I.e., Carbon always has 6 protons. So, this isotope is also written: 12 C or Carbon-12 C 12 6
- 29. In chemical reactions , these items do not change Atomic number (Z) is constant Mass number (A) is constant Element does not change If any of these values change, then it is a nuclear reaction! Transmutation of an Element C 12 6
- 30. Examples of nuclear reactions U 238 92 Th 234 90 He 4 2 (Plus a lot of kinetic energy) Alpha-decay n 1 0 N 14 7 Neutron capture p 1 1 C 14 6 (This reaction happens in the upper atmosphere to produce Carbon-14)
- 33. Let’s understand beta-decay a little more by learning a bit about particle physics
