DVT prophylaxis and Pradax
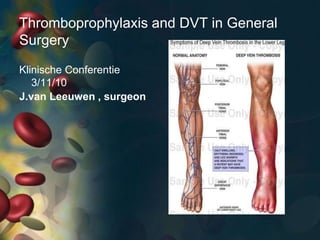
- 1. Thromboprophylaxis and DVT in General Surgery J.van Leeuwen , surgeon Klinische Conferentie 3/11/10
- 2. We should be aware that . . . • 70% of all VTE is hospital-acquired • VTE is the commonest preventable cause of hospital death • Thromboprophylaxis the number 1 ranked patient safety strategy in hospitalized patients
- 3. As general surgeons or clinicians . . . • What can you do to reduce the burden of VTE? • Be knowledgeable about VTE • Assess your patients for VTE risk and use thromboprophylaxis routinely • Support hospital-wide thromboprophylaxis
- 4. DVT Blood Clots: A Potentially Fatal Health Problem • Up to 2 million people in the United States suffer from DVT blood clots every year • Approximately 600,000 experience pulmonary embolism (PE) • In the US, complications from DVT blood clots kill almost 300,000 people a year — more than AIDS and breast cancer combined
- 5. DVT •Common, potentially life – threatening • medical problem •Is better prevented but once happened should • by aggressively treated •Risk factors (Virchow’s Triad) •50% of DVT are “silent”
- 6. DVT Swelling of the leg Pain or tenderness in the leg; the pain is usually in 1 leg and may only be present when standing or walking Leg feels warm to the touch Red or discolored skin
- 7. PE Unexplained shortness of breath Chest pain or palpitations Anxiety and/or sweating Coughing up blood Fatigue and/or fainting
- 8. Some of the risk factors that may increase risk of DVT • Age 40 years or older • Being overweight • A personal or family history of blood clots • Birth control pills • Hormone replacement therapy (HRT) • Cancer
- 9. • Certain heart problems • Respiratory failure • Varicose veins • Pregnancy • Surgery, especially hip, knee, or abdominal surgery • Currently have restricted mobility due to a long illness or surgery
- 12. DVT Prophylaxis Early ambulation Stockings with or without pneumatic compression Antiplatelets Anticoagulant (low dose heparin)
- 13. Indications For Antithrombotic Therapy • Venous thromboembolic disease • Deep venous thrombosis (DVT) • Pulmonary embolism (PE) • Arterial thromboembolic disease • Prosthetic heart valves • Mitral valve disease • Congestive cardiomyopathies • Atrial fibrillation • Mural cardiac thrombi • Transient ischemic attacks • Stroke in evolution Disseminated intravascular coagulation • Maintenance of patency of vascular grafts, shunts, bypasses
- 15. Practical measures Elevate the affected leg whenever possible • Apply heat to relieve pain and reduce swelling • Wear compression bandages or support hose • Avoid long periods of immobility
- 16. Treating DVT • Treatment of DVT can help reduce complications such as PE • The main goals in treating DVT are to: • Stop the clot from getting larger • Reduce the chance of developing another clot • Reduce the risk of the clot breaking off in your vein and moving to your lungs
- 17. Complications of DVT PE Secondary varicose veins Postphlebitic limb: Varicose veins, ulcer, pain, change of colour, lipodermatosclerosis
- 18. Treatment of DVT Legs elevation iv. heparin infusion and monitoring by PTT acenocoumarol and monitoring by INR LMWH, fibrinolytic Rx I.V.C. filter Surgical thrombectomy
- 19. Anti Platelet Drugs Drug Mechanism Uses Aspirin Permanently inhibits COX-1 and COX-2 CAD Stroke-TIAs NSAIDs Reversibly inhibits COX-1 Limited Dipyridamole Inhibits PDE; increases cAMP TIAs Ticlopidine Clopidrgrel Inhibits ADP PlatAg;active metabolite TIAs;Stroke CAD;PVD
- 20. Platelet Receptor Mediated Pathways: Drugs Arachidonic Acid ASA NSAIDs ADP Ticlopidine Clopidogrel Thrombin -Final Common Pathway -Promotes Platelet Adhesion (Fibrinogen, vWF) GP IIB/IIIA Inhibitors Abciximab (ReoPro) Eptifibatide (Integrilin) Tirofiban
- 21. Contraindications to Antithrombotic Therapy -Recent thoracic, abdominal, or central nervous system surgery -Recent cerebrovascular accident, trauma, or neoplasm -Bleeding ulcer -Hypertension -Anticipated invasive procedures (arterial punctures, biopsies, central lines) -Concurrent hemostatic dysfunction
- 22. Contraindications to Antithrombotic Therapy • Specific to acenocoumarol (ambulatory patients) -Early and late pregnancy -Poor patient cooperation, understanding, reliability -Unsatisfactory laboratory or patient follow-up -Occupational risk to trauma
- 23. Contraindications to Antithrombotic Therapy • General risk factors -Pre-existing coagulation or platelet defect, thrombocytopenia, or other bleeding abnormality -Inaccessible ulcerative lesion (e.g., gastrointestinal tract lesion) -Central nervous system lesion (e.g., caused by stroke, surgery, trauma) -Spinal anesthesia or lumbar puncture -Malignant hypertension -Bacterial endocarditis -Advanced retinopathy -Old age (relative) -Aspirin or other antiplatelet drugs -Neoplastic disease
- 30. Low Dose Unfractionated Heparin • Surgical Prophylaxis • 5,000 Units SQ 2 hr preop • 5,000 Units SQ every 12 hours • Medical Prophylaxis • 5,000 Units SQ every 12 hours • No monitoring required
- 31. Monitoring of Anticoagulant Therapy Heparin s.q. – no monitoring required i.v. - partial thromboplastin time (P.T.T.) mechanism – measures intrinsic pathway therapeutic goal – 2-2.5 times normal control value (-30 sec)
- 32. Unfractionated Heparin • High Dose • Treatment of venous/arterial thrombi • Requires monitoring • IV- 5,000 Units bolus, then 30,000- 35,000 units/24 hrs • 80 Units/kg bolus, then 18 Units/kg/hr to maintain aPTT in therapeutic range
- 33. Dabigatran etexilate is a novel medicine, small molecule, reversible, direct thrombin inhibitor For oral administration the prodrug dabigatran etexilate was developed in 75 and 110 mg NH2 N NH N N CH3 N OO O N O O CH3 CH3 Dabigatran etexilate What is Pradaxa?
- 34. • Oral prodrug, converted to dabigatran, a potent reversible direct thrombin inhibitor (DTI) • Half life of 12-17 h, • ~ 80% renally excreted • 6.5% bioavailability • Rapid onset of action • Predictable and consistent anticoagulant effects • Low potential for drug-drug interactions, no drug-food interactions • No requirement for routine coagulation monitoring • Potent antithrombotic effects are achieved with direct thrombin inhibitors by specifically blocking the activity of thrombin (both free and clot- bound), the central enzyme in the process responsible for clot (thrombus) formation Dabigatran etexilate:
- 35. What is Pradaxa used for? • Pradaxa is used to prevent the formation of blood clots in the veins in adults who have had an operation to replace a hip or knee , or any operation with risk of forming clots.
- 36. How is Pradaxa used? • Start with one 110 mg capsule taken one to four hours after the end of the operation. Treatment then continues with two 110 mg capsules (220 mg) once a day for 28 to 35 days after hip replacement and for 10 days after knee replacement. • A lower dose (150 mg once a day) is used in patients with mild or moderate kidney problems
- 37. How does Pradaxa work? • The active substance in Pradaxa, dabigatran etexilate, is a ‘prodrug’ of dabigatran. This means that it is converted into dabigatran in the body. Dabigatran is an anticoagulant, meaning that it prevents the blood from coagulating (clotting). It blocks thrombin, which is central to the process of blood clotting, reducing the risk of blood clots forming in the vein
- 38. How has Pradaxa been studied? • The effectiveness of Pradaxa was studied in three main studies, both of which compared Pradaxa with enoxaparin (another anticoagulant). • Orthopedic surgery study and Re-LY study in the efficacy of this drug for AF
- 39. The RE-LY Study: Randomized Evaluation of Long-term anticoagulant therapY Dabigatran Compared to Warfarin in 18,113 Patients with Atrial Fibrillation at Risk of Stroke Connolly SJ., et al. NEJM published online on Aug 30th 2009. DOI 10.1056/NEJMoa0905561 Dabigatran etexilate is in clinical development and not licensed for clinical use in stroke prevention for patients with atrial fibrillation
- 40. RE-LY® – summary results versus warfarin • Statistically significant reduction in stroke/systemic embolism • Statistically significant reduction in hemorrhagic stroke • Statistically significant reduction in vascular mortality • Comparable rates of major bleeding rates • Significant reduction in total bleeds, life threatening bleeds and intracranial bleeds Connolly SJ., et al. NEJM published online on Aug 30th 2009. DOI 10.1056/NEJMoa0905561
- 41. • The other studies involved a total of 2,101 patients who had had a knee replacement operation, and • The second involved a total of 3,494 patients who had had a hip replacement.
- 42. • In these studies, the main measure of effectiveness was the number of patients who formed blood clots in the veins or who died of any cause during the treatment period. In most cases, blood clot formation was detected using scans of the veins or by looking for signs of blood clots in the lungs.
- 43. What benefit has Pradaxa shown during the studies? • In both studies, Pradaxa was as effective as enoxaparin in preventing the formation of blood clots or death. • In the study of patients undergoing knee replacement, blood clots were detected in 182 (36%) of the 503 patients taking Pradaxa, compared with 192 (38%) of the 512 receiving enoxaparin.
- 44. • There was only one death in each group (less than 1%). After hip replacement, blood clots were detected in 50 (6%) of the 880 patients taking Pradaxa, compared with 60 (7%) of the 897 receiving enoxaparin. • Three patients in the Pradaxa group died (less than 1%), but two of these deaths were unrelated to blood clots.
- 45. • After hip replacement, blood clots were detected in 50 (6%) of the 880 patients taking Pradaxa, compared with 60 (7%) of the 897 receiving enoxaparin. • Three patients in the Pradaxa group died (less than 1%), but two of these deaths were unrelated to blood clots.
- 46. What is the risk associated with Pradaxa? • The most common side effect with Pradaxa (seen in more than 1 patient in 10) is bleeding. • Pradaxa should not be used in people who may be hypersensitive (allergic) to dabigatran etexilate or any of the other ingredients. • Not use in patients who have severe problems with kidneys, active significant bleeding, tissue damage that could lead to bleeding, problems with the blood clotting process .
