Electron Configuration
•Transferir como PPT, PDF•
18 gostaram•9,527 visualizações
The document summarizes the evolution of atomic models from Dalton's billiard ball model in 1803 to the modern quantum mechanical model. It traces the key discoveries and models of Thomson, Rutherford, Bohr, Schrodinger that led to the understanding that electrons occupy discrete energy levels and orbitals around the nucleus rather than definite orbits, described by quantum numbers. The modern atomic model uses orbitals and quantum mechanics to describe the probability of finding electrons in an atom.
Denunciar
Compartilhar
Denunciar
Compartilhar
Recomendados
Mais conteúdo relacionado
Mais procurados
Mais procurados (20)
Chemistry - Chp 5 - Electrons In Atoms - Powerpoint

Chemistry - Chp 5 - Electrons In Atoms - Powerpoint
Destaque
Destaque (20)
Chapter 5.3 : Electron Configuration and the Periodic Table

Chapter 5.3 : Electron Configuration and the Periodic Table
Semelhante a Electron Configuration
Semelhante a Electron Configuration (20)
Último
Último (20)
Passkey Providers and Enabling Portability: FIDO Paris Seminar.pptx

Passkey Providers and Enabling Portability: FIDO Paris Seminar.pptx
Testing tools and AI - ideas what to try with some tool examples

Testing tools and AI - ideas what to try with some tool examples
Long journey of Ruby standard library at RubyConf AU 2024

Long journey of Ruby standard library at RubyConf AU 2024
So einfach geht modernes Roaming fuer Notes und Nomad.pdf

So einfach geht modernes Roaming fuer Notes und Nomad.pdf
Potential of AI (Generative AI) in Business: Learnings and Insights

Potential of AI (Generative AI) in Business: Learnings and Insights
(How to Program) Paul Deitel, Harvey Deitel-Java How to Program, Early Object...

(How to Program) Paul Deitel, Harvey Deitel-Java How to Program, Early Object...
The Fit for Passkeys for Employee and Consumer Sign-ins: FIDO Paris Seminar.pptx

The Fit for Passkeys for Employee and Consumer Sign-ins: FIDO Paris Seminar.pptx
Take control of your SAP testing with UiPath Test Suite

Take control of your SAP testing with UiPath Test Suite
A Journey Into the Emotions of Software Developers

A Journey Into the Emotions of Software Developers
Modern Roaming for Notes and Nomad – Cheaper Faster Better Stronger

Modern Roaming for Notes and Nomad – Cheaper Faster Better Stronger
Time Series Foundation Models - current state and future directions

Time Series Foundation Models - current state and future directions
UiPath Community: Communication Mining from Zero to Hero

UiPath Community: Communication Mining from Zero to Hero
The Future Roadmap for the Composable Data Stack - Wes McKinney - Data Counci...

The Future Roadmap for the Composable Data Stack - Wes McKinney - Data Counci...
Assure Ecommerce and Retail Operations Uptime with ThousandEyes

Assure Ecommerce and Retail Operations Uptime with ThousandEyes
Electron Configuration
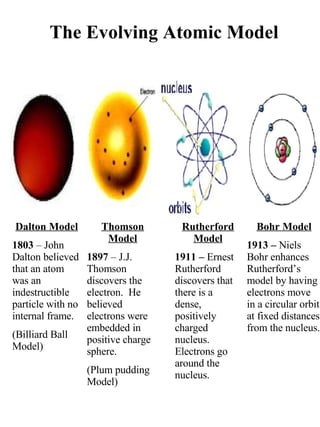
- 1. Dalton Model 1803 – John Dalton believed that an atom was an indestructible particle with no internal frame. (Billiard Ball Model) Thomson Model 1897 – J.J. Thomson discovers the electron. He believed electrons were embedded in positive charge sphere. (Plum pudding Model) Rutherford Model 1911 – Ernest Rutherford discovers that there is a dense, positively charged nucleus. Electrons go around the nucleus. Bohr Model 1913 – Niels Bohr enhances Rutherford’s model by having electrons move in a circular orbit at fixed distances from the nucleus. The Evolving Atomic Model
- 13. Study guide for chp. 4 and 5 Atomic structure notes -Know how to calculate atomic mass -Know how to draw bohr model Review worksheets 4.1-4.3 Know what ion an element will form Quantum mechanics notes Know how to write long hand electron configuration Be able to explain the behavior of electrons (i.e., what did you learn from the flame lab?) Review worksheets 5.1-5.2
- 14. Need to add Relate outer shell to lewis dot and oxidation states