The Distillation of Essential Oils Part 2
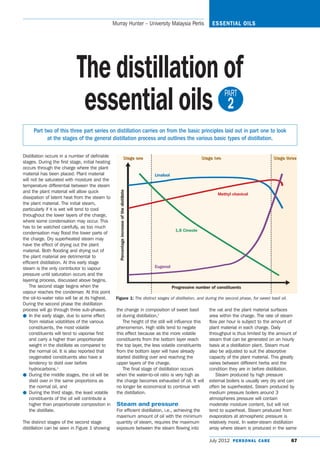
- 1. Murray Hunter – University Malaysia Perlis ESSENTIAL OILS The distillation of essential oils 2: PART Part two of this three part series on distillation carries on from the basic principles laid out in part one to look at the stages of the general distillation process and outlines the various basic types of distillation. Distillation occurs in a number of definable Stage one Stage two Stage three stages. During the first stage, initial heating occurs through the charge where the plant material has been placed. Plant material Linalool will not be saturated with moisture and the temperature differential between the steam Percentage increase of the distillate and the plant material will allow quick Methyl chavicol dissipation of latent heat from the steam to the plant material. The initial steam, particularly if it is wet will tend to cool throughout the lower layers of the charge, where some condensation may occur. This has to be watched carefully, as too much 1,8 Cineole condensation may flood the lower parts of the charge. Dry superheated steam may have the effect of drying out the plant material. Both flooding and drying out of the plant material are detrimental to efficient distillation. At this early stage steam is the only contributor to vapour Eugenol pressure until saturation occurs and the layering process, discussed above begins. The second stage begins when the Progressive number of constituents vapour reaches the condenser. At this point the oil-to-water ratio will be at its highest. Figure 1: The distinct stages of distillation, and during the second phase, for sweet basil oil. During the second phase the distillation process will go through three sub-phases. the change in composition of sweet basil the vat and the plant material surfaces ɀ In the early stage, due to some effect oil during distillation.2 area within the charge. The rate of steam from relative volatilities of the various The height of the still will influence this flow per hour is subject to the amount of constituents, the most volatile phenomenon. High stills tend to negate plant material in each charge. Daily constituents will tend to vaporise first this effect because as the more volatile throughput is thus limited by the amount of and carry a higher than proportionate constituents from the bottom layer reach steam that can be generated on an hourly weight in the distillate as compared to the top layer, the less volatile constituents basis at a distillation plant. Steam must the normal oil. It is also reported that from the bottom layer will have already also be adjusted to suit the absorptive oxygenated constituents also have a started distilling over and reaching the capacity of the plant material. This greatly tendency to distil over before upper layers of the charge. varies between different herbs and the hydrocarbons.1 The final stage of distillation occurs condition they are in before distillation. ɀ During the middle stages, the oil will be when the water-to-oil ratio is very high as Steam produced by high pressure distil over in the same proportions as the charge becomes exhausted of oil. It will external boilers is usually very dry and can the normal oil, and no longer be economical to continue with often be superheated. Steam produced by ɀ During the third stage, the least volatile the distillation. medium pressure boilers around 3 constituents of the oil will contribute a atmospheres pressure will contain higher than proportionate composition in Steam and pressure moderate moisture content, but will not the distillate. For efficient distillation, i.e., achieving the tend to superheat. Steam produced from maximum amount of oil with the minimum evaporators at atmospheric pressure is The distinct stages of the second stage quantity of steam, requires the maximum relatively moist. In water-steam distillation distillation can be seen in Figure 1 showing exposure between the steam flowing into array where steam is produced in the same July 2012 P E R S O N A L C A R E 67
- 2. ESSENTIAL OILS vessel that the plant material is stored will that the temperature gradient increases, Recently a variation on the operation of produce very wet steam. thus increasing the effects of latent heat pressure in steam distillation has been Different steams can be utilised as a exchange and reduces the likelihood of reported a number of times, utilising a control mechanism to correct the hydrolysis. However the use of high technique called instantaneous controlled conditions within the still housing the plant pressure distillation is limited by the extent pressure drop (DIC). This is a technique charge. that prolonged high temperatures will where the foliage is first exposed to Wet steam is a saturated vapour is damage the composition of the essential saturated steam and then the pressure suitable for most distillation. In most cases, oil. drastically dropped to a vacuum level of wet steam from a water bath produces ‘a Inversely, as operating pressure is around 5-50 KPa to provoke auto- richer oil’ with much shorter extraction time reduced, so does the temperature of the vaporisation of the superheated volatile than other forms of steam.3 This is distillation. This method can be used for compounds through expanding and particularly the case with plant material the extraction of heat sensitive constituents breaking up the cell walls with that contains superficial oil glands. that would normally be damaged through instantaneous cooling.4 Experiments have Increasing steam rates does not speed up exposure to excess heat. However shown that results can be varied through distillation, especially if the process of distillation under reduced pressure has a changing time, pressures and the amount hydro-diffusion is required. As plant number of limitations. Steam under of moisture in the leaves.5 material is already saturated with moisture reduced pressure is less dense, so requires and there is a constant diffusion rate. If more steam to carry out a distillation than Wilting crops before herbs contain a lot of moisture, then there under normal atmospheric pressure. The distillation would be sufficient moisture in the plant condenser system would need to be As the moisture condition of the herb is a material to commence hydro-diffusion and almost twice the size of a conventional still factor in the efficient distillation of the dry steam would be the most suitable to or a refrigeration system required in herb, wilting is often carried out to dry the apply. However dry steam has less mass condensing the distillate. The recovery crop before processing. The objective of than wet steam and as such reduces the vessel and separator would have to be wilting is to dry the herb enough to latent heat of steam, thus prolonging the sealed within the closed system, which increase its absorptive surface. Many distillation period. would lead to design and engineering practitioners believe it is to dry out excess A superheated steam occurs when the difficulties. moisture so that distillation will be shorter temperature of the vapour is higher than that of the same saturated vapour at the same pressure. Superheated steam is independent of pressure and therefore advantageous in a number of situations, as it can be utilised at any temperature without increasing pressure, i.e., can increase the temperature of distillation without having to change the steam flow- rate due to changes in pressure. Superheated steam can be used for drying out a flooded charge vessel, where there is too much liquid around the plant material. However superheated steam is not advisable for general distillation as it would dry out the plant material, preventing the hydro-diffusion process occurring and is a poor conductor of latent heat – two of the important processes needed for efficient distillation. Superheated steam is most suitable for the distillation of glabrous herb material and can increase yields substantially. High boiling oils exert less vapour pressures and require relatively large temperature gradients to extract them during distillation. This leads to prolonged distillation periods. If the constituents are stable under long periods of heat, distillation can be performed under pressures above the atmosphere, which increases temperature. This cuts back on distillation time and saves energy. As increasing the still pressure, increases temperature, the temperature gradient will also increase between the vapour space in the still and the plant material. This assists in the vaporisation of high boiling volatile Figure 2: Mint foliage, cut and laid on the ground for wilting, being picked up by the harvester constituents. The advantage of pressure is for distillation at Myrtlefor, Victoria. 68 P E R S O N A L C A R E July 2012
- 3. ESSENTIAL OILS and more efficient. However this is a Condenser cooling fallacy. For example, with tough leaves like water outlet eucalyptus and tea tree, wilting does little to dry them out or increase their surface Vapour condenser absorption, so wilting will have no benefit to the distillation process. Moisture in the Vapour outlet plant material is actually of benefit to the to condenser distillation process.6 In fact there are risks with wilting in that the process may lead to Condenser cooling Still chamber for water inlet losses in oil, not through evaporation, but plant material through chemical reactions like oxidisation, (loaded by resinisation and the formation of glycosides cartridge, and enzymes in the materials. Excessive or directly) drying of moisture can remove necessary moisture breaking the contact between the Welded wire Separator oil component and surface of the plant Steam inlet mesh floor flask material, thus hindering the promotion of from boiler Steam outlet hydro-diffusion during the distillation process. Which crops require wilting before Still drain valve distillation generally depends upon their natural surface absorption capacity. Non- Figure 3: Diagram of a steam distillation still. absorptive herbs like mint and basil need some wilting to promote absorption during which need to be comminuted before become part of the boiling mixture and distillation, as wilting to promote partial distillation, like almond powder and huon resulting mixed vapour. breakdown of the surface cellular structure pine saw dust. As water boils and converts to steam at of the leaf. In this case distillation would In water distillation there are a number the bottom of the vat and rises through the then commence with a wet steam fraction, of simultaneous processes that act to plant charge, it will come into contact with followed by slightly drier steam once extract volatile constituents from plant the plant material. Some oil is exposed on moisture has permeated into the leaf material that are different from steam the surface of this material will be structure. Herbs with absorptive surfaces distillation. vaporised by the rising steam as it comes like lavender do not need to be wilted. In Essential oils contain a number of into contact with the plant surfaces. This fact they do not have enough moisture to oxygenated constituents that are relatively steam carrying some volatile vapour will link the oil glands to the surface via a water soluble in water. This would include rise to the surface and carry over into the interface, so wet steam fractions are phenols, alcohols and some aldehydes. vapour space above the water until it needed during distillation. Grass crops like During the early stages of a water reaches the still condenser. lemongrass and citronella contain enough distillation, these compounds would The boiling temperature of water at the moisture within their leaf structure to dissolve in the surrounding water and bottom of the vat in water distillation is create a water-oil-surface interface for hydro-diffusion to occur during distillation, Table 1: Some common water soluble aroma chemicals in essential oil. so wilting is not necessary. As moisture content is already sufficient in the leaf, dry Slightly soluble Moderately soluble Very soluble steam would be suitable for the distillation (<500 ppm) (501-1999 ppm) (>2000 ppm) process. Aldehyde C11 Calamene Benzlaldehyde Aldehyde C12 (lauric) 1,8-Cineole Benzyl acetate Water distillation Aldehyde C12 MNA Nerol Cinnamic alcohol Water distillation involves distilling plant material totally immersed in water. Amyl cinnamic aldehyde Neryl acetate Citronellyl acetate Depending upon the specific gravity and Amyl salicylate Rose oxide α-Copanene charge mass in the still, the material will Benzyl salicylate Eugenol either float or sit totally immersed in the Cedrol Geranial water. Heat is introduced by direct heating Citronellol Geraniol of the sides of the vat, a steam jacket, a Citronellyl butyrate Limonene closed system coil or in some cases a perforated steam coil. Water distillation was Citronellyl formate Linalool the only method used before the 20th Eudesmol Mentone century. Geranyl formate Myrcene Water distillation is useful for the Limonene (E)-β-ocimene distillation of flower materials which would Linalyl acetate Phenylethylacetate normally congeal and form lumpy masses α-Pinene Phenylethylalcohol under steam distillation, where steam would not penetrate, like rose petals and Sabinene orange blossoms. This method is also Terpinen-4-ol useful for fruit kernels that would form α-Terpineol glutinous masses under steam distillation Terpinolene and powdered forms of plant material July 2012 P E R S O N A L C A R E 69
- 4. ESSENTIAL OILS slightly less than the boiling point of water, the plant surface material as possible material from directly coming into contact due to the mixed liquid of solublised during the distillation. with the heating coils. volatiles and water. Heat applied to the still The effects of hydro-diffusion are much With many materials, part of the oil will cause the creation of a small bubble of slower in water than other types of dissolves in the water during distillation and saturated mixed vapour from the liquid distillation. Consequently, especially for forms a milky emulsion, as a number of phase, where upon formation it rises to the wood materials extensive comminution aromatic constituents are soluble to some top of the water in the charge. During the must be undertaken so that particles in the degree in water. This loss could range up to rise, the bubble’s pressure, temperature charge are fine and as much oil as possible 25% of the essential oil.7 This means that and proportion of oil to water decreases. is exposed on the surface of the material. the recovery of oil is incomplete and the The condensing volatiles, mostly being less In water distillation, plant material is recovered oil will be deficient in some dense than water, float to the top of the placed in a sealed vessel or retort that constituents that would be the case with water and form a film on the surface of the connects directly to a condenser. From the the oil recovered through steam distillation. water in the vessel. This lost oil tends to condenser the distillate runs into a Upon separation in water distillation, the remain on top of the surface and cannot separator. The rate of distillation is water distillate is returned directly to the re-vaporise easily due to its higher boiling controlled by the intensity of the fire, the charge vessel to replace the decreasing point and the generally cooler temperatures pressure of the vessel or retort and/or the water level due to evaporation. This is at the water surface. Most of the oil rate of introduction of steam. As many called cohobation. Sometimes the water recovered in water distillation is the portion woods contain high boiling compounds, distillate is redistilled in another vessel to of the oil that does not condensate through pressure is vital to create high enough extract the volatiles in emulsion. Salt is this action. temperatures to vaporise the volatile often added to the distillate to reduce the Observation shows that distillation constituents. These constituents may take solubility of water. Whether this process is undertaken with vigorous boiling produces many hours to boil out. Hydro-stills should undertaken depends upon the probability of better and quicker yields than mild boiling. generally be wide to maximise the the constituents being damaged by further Some distillers even install small propellers evaporation area. Where particles are fine heat and the economics of re-distillation. intruding into the side of the still to assist such as saw dust and powders some form Another method to recover the dissolved in agitation. This is most probably effective of mesh or “P” shape pipe arrangement aromatic materials from the water distillate because the agitation in the charge tank should exist at the entrance to the is to add a solvent. The mixture is then prevents oil droplets clinging to the herb condenser to prevent plant material from vigorously shaken to pick up dissolved surfaces. It is necessary to generate entry and possible clogging. Heavy charges constituents from the water into the enough steam in the water so that it will and where heat coils are used in the still solvent. These materials are then recovered come in contact with as much of the oil on require a perforated grid to prevent plant through vacuum distillation of the solvent Table 2: A comparison between water, water and steam and steam distillation. Water distillation Water and steam distillation Steam distillation Construction Usually simply constructed, Inside vat contains a grill where Utilises an outside steam source based on ancient designs. plant material rests above the water Plant material suitability Finely powdered materials such Can be used for most herb and Most materials, except fine as woods and rhizomes, flowers leaf materials. Material must powders. Good for high boiling that congeal with steam. be packed in a uniform materials. Material must be packed Not suitable for materials that manner to avoid channeling in a uniform manner to contain acidic material which can of steam. avoid channeling of steam. saponify, water soluble or high boiling constituents. Material must be completely covered with water. Hydro-diffusion Excellent Good Steam should be slightly wet to promote diffusion. Superheated or high pressure steam can dry out the plant material and inhibit diffusion. Available Pressure Atmospheric pressure Atmospheric pressure Can be adjusted for both high and reduced pressures. Available Temperature 100˚C, but care must be taken Approximately 100˚C Can be increased or reduced not to burn the plant material according to pressure used. from direct heat on the sides. Hydrolysis Condition Usually a high rate of ester Hydrolysis is usually low, however Slight hydrolysis. hydrolysis. excessive wetting of the plant material through prolonged distillation can promote hydrolysis during later stages of distillation. Rate of distillation and yield Slow rate of distillation. Moderate distillation rate. Fast rate of distillation. Low rates of yield due to Good yields if no channels Good yields if no channels hydrolysis and loss of water occur in charge. Usually good yields. occur in charge. Usually good yields. soluble constituents into the water. High boiling constituents often left undistilled. 70 P E R S O N A L C A R E July 2012
- 5. ESSENTIAL OILS which results in a secondary essential oil.8 Some common water soluble aroma chemicals in essential oils are listed in Table 1. Another method that will contribute to minimising oil loss due to oil solubility in water during the separation phase is to control the outgoing distillate temperature from the condenser. Where oils are less dense than water, there will be an optimal temperature range where oil particles will freely float to the top of the distillate upon condensation. Some literature on distillation practices misses the point about the effect of condensation temperatures on oil yields.9 Based on private work,10 the higher the temperature of the outgoing distillate, the freer will be the oil particles to float to the top. For example, tea tree oil droplets will float to the top of the water distillate twice as fast at 60°C than at 40°C. The upper temperature limit will be restricted by the potential loss of low boiling volatiles during condensation. This has implications on the design of the Figure 4: The condenser, with the end manifold removed to expose the internal cooling pipes, specific condenser for specific crops and at the author’s property in Perlis, Malaysia. set range limits upon the temperature that distillation can take place, to achieve a water distillation. Water and steam steam distillation facilities are designed and specific outgoing distillate temperature distillation has another advantage over built with the correct steam ratings, they range. steam distillation as there are fewer are much more economical to run than decomposed products during the process water and water and steam distillation Water and steam distillation due to less chance of plant material drying systems. A comparison between water, Water and steam distillation involves the out. However water and steam distillation water and steam and steam distillation is storing of the plant material above a water will take a lot longer. Water and steam shown in Table 2. below. bath situated in the bottom of the charge distillation can produce very good results vessel and heating the water either through under reduced pressure. The condenser system direct fire, a steam jacket or a closed or Water and steam distillation is much A condenser in an essential oil distillation open steam coil. Water and steam cheaper to set up than steam distillation system is a heat exchange or dissipating distillation produces saturated wet steam at facilities and lends itself to portable stills device. The condenser must not only cool the prevailing vessel pressure, which is that can be transported from place to the condensate vapour into a liquid, but usually atmospheric pressure. Within this place. also cool the condensate to the configuration, unlike water distillation, only temperature range where the oil will steam comes in contact with the plant Steam distillation separate spontaneously from the water. material. Steam distillation employs an external The usual array for a condenser system Water and steam distillation is not very steam generation system, external to the is a tube or set of tubes running through a suitable for fine materials as steam will find charge vessel. This configuration provides sealed water reservoir to cool the pipes. It a path of least resistance tending to create much more control (depending upon the is immediately attached to the top of the channels. This means that steam will not boiler capacity) than water and steam charge vessel to collect all vapour exiting flow through the whole mass of plant distillation. This is because in steam the vessel. The design of the condenser material and an incomplete distillation will distillation the wetness fraction, must ensure that the vapour flow is take place. If the plant material is loosely temperature and pressure can be turbulent inside the tubes to prevent high packed, the same effect will happen, as manipulated according to needs and velocity vapour freely flowing through the the material will offer no resistance to the conditions. However, it is a misconception condenser and maximise exposure to the steam. that greater steam volumes and increased cooler walls. A baffle is usually installed at Water and steam distillation may take a pressures have positive effects on the the beginning of the condenser to disrupt a long period of time to reach operating process in all cases. As mentioned straight steam flow for this purpose. Failure temperature as the plant material needs to previously, dry and superheated steam has to achieve those conditions would result in be heated up with only saturated steam. the effect of drying out plant material, some vapour failing to condensate. Within This may cause early condensation and which potentially halts distillation through the condenser system the flows of vapour wetting of the plant material. the stopping of hydro-diffusion process. and cooling water should be in opposite Due to the limits on pressure that can Faster steam flow rates do not necessary directions at the maximum possible speed. be built up in the charge vessel, water and mean quicker recovery times. Fabricated The condenser must be sensitive enough to steam distillation will have only a limited steam boilers cost much more to run than react on the vapour flow very quickly. The effect on extracting high boiling materials water baths and may lead to high required number of tubes and length of the from plant materials. However there is less distillation costs, especially if they require condenser depends on the rate of distillate opportunity for hydrolysis to occur than with petrochemical feed stocks. However if flow, the pressure, the temperature of the July 2012 P E R S O N A L C A R E 71
- 6. ESSENTIAL OILS cooling water and the desired exit microscopic oil droplets. Temperature plays Distillate in-flow from still temperature range of the distillate. (water-oil mixture) a crucial role in separation, where it should The condenser must remove the be moderately warm to increase the equivalent amount of heat that is needed relative specific gravity differential of the oil to vaporise the distillate, plus the additional Oil and water. Raised temperature of the outlet amount of heat to reach the optimal distillate water will allow the small oil distillate temperature range of the particles to rise to the top of the separator condensate distillate exiting the condenser. quicker in a similar manner to the The rate of which heat would be removed condenser exit temperature range where from the distillate can be represented by Water there will be an optimal separator water Internal outlet the following equation: baffle temperature range to promote oil-water Q = UA∆t ∆ separation.11 Where Q = the heat removed by unit During distillation, the more volatile of time constituents tend to vaporise quicker and U = a constant determined by the less volatile constituents vaporise later operating conditions (condensing and in the distillation. This leads to an oil that cooling made up of a number of will vary in constituents during the factors) usually a constant is used. distillation period. By changing separation A = the area available for heat flasks at particular points during a single removal distillation, oils of different constituent ∆t = the temperature difference profiles can be collected. This is important between the vapour and the cooling Figure 5: Schematic view of a hydro-diffusion in ylang ylang and lavender distillation, medium. distillation system. where different oils profiles will have different uses and values to particular U is made up of a number of factors possible separate the oil from the distillate customers. Many distillers also use this including the flow rates of cooling water water. As distillate water volume is much principal to collect specific fractions during and vapours, the material that the greater than oil, it is important that water the distillation, which can later be blended condenser is constructed, and usually a can be removed continuously. Oil and together to create a whole oil that meets constant is used due to the difficulty to water separates according to specific with certain specific specifications, such as calculate. The value of U increases as gravity forming two layers. Lighter than a standard. these factors increase. Thus according to water oils will float to the top and heavier The water distillate will always be the equation, the surface area can be as than water oils will sink to the bottom. This saturated in oil and directly dumping it large or small as desired, as long as the must be considered in separator design for would lead to a loss in yield. For this other factors compensate. However the water removal. reason some distillers (water and water and overall capacity of the distillation system If the specific gravity of oil and water is steam distillation) will channel the water will have great bearing on the condenser very close, the two components will not distillate back into the still vessel for re- area. Condenser sizes will also vary in size separate immediately. Distillate flowing into distillation in what is called cohobation, according to the temperature of available the separator must therefore not disturb mentioned previously. For this purpose the cooling water on site, thus condensers in the surface area and flow into the body of separator must be placed higher than the temperate and tropical areas will reflect the water to prevent surface turbulence. still vessel proper so distillate water in the this in size. The separator must also be large enough separator can be fed back into the still Using a condenser system with the so that drained water does not carry away through gravity. wrong capacity for the distillation system will have a number of operational Steam inlet consequences. A too efficient condenser system will deliver the distillate at a temperature below the optimum range, which could lead to cool air outside being Downward steam flow sucked into the system. This outside cooler air in the condenser tubes could create expansion and contraction of vapours in Plant material the condenser leading to splattering and Side opens so that intermittent distillate outflow. This could cartridge with plant also occur if the cooling water is too cold. material can slide in Grated base If the condenser is too small for the and out of still distillation system, then the still must be operated with lower steam rates, which Condenser array would lengthen distillation times and open up the possibility of hydrolysis to occur within the vessel. Water and oil collector The separator system Before leaving the subject of distillation in this chapter, some words about the Collector vessel separator system must be mentioned. The function of the separator is to as quickly as Figure 6: Oil separator design used in Perlis, Malaysia. 72 P E R S O N A L C A R E July 2012
- 7. ESSENTIAL OILS Hydro-diffusion material near the bottom may also lead to 6 Guenther E.The Essential Oils, Volume One: distillation hydrolysis and lead to lesser yields. History – Origin in Plants, Introduction – Analysis, Hydro-diffusion distillation is a variation on However, hydro-diffusion distillation appears Malabar, Florida, Robert E. Krieger Publishing steam distillation where steam is popular within the aromatherapy industry in Company, P 110, 1948. . introduced on the top of the vessel and Europe. PC 7 Fleisher A. Water-soluble fractions of the condenses through the plant material in essential oils, Perfumer & Flavorist 1991; the still, where the distillate is collected ɀ The final article in September will 16 (3): 37-41. and condensed under the plant material conclude with a brief discussion about 8 Bouzid N, Toulgouate K, Villarem G, Gaset A. which rests on a grill or perforated tray. applying these principles. Analyse quantitative des fractions d’huile Through steam travelling down the still, essentielle pouvant co-exister lors there is more time for the volatiles and References d’hydrodistillation de plants aromatiques, fatty acids floating on the plant material. 1 Johannes AK, Scheffer JC, Svendsen AB. Rivista Ital Eppos 1997; 79: 15-25. In the case of wood and seeds that have Comparison of Isolation Procedures for Essential 9 Boland DJ, Brophy JJ, House APN. Eucalyptus many high boiling compounds, which are Oils, Z. Lebensm. Unters. Forsch, Vol. 168, Leaf Oils: Use, Chemistry, Distillation and difficult to vaporize in an ordinary still, this 1979, pp. 106-111. Marketing 1991; Melbourne, Inkata Press, P . system may be effective. This would be 2 Private communication with Mr. Jon Bonnardeaux 191. valuable when fatty acids contribute to the of Western Australia Department of Agriculture, 10 Unpublished private work on tea tree distillation flavour of a material and it is desirable in early 1990s. at Batu 9, Berseri, Perlis, Malaysia during the oil. Thus, hydro-diffusion distillation 3 Denny EFK. Field Distillation for Herbaceous Oils, 2004/5. may return an oil more representative of Lilydale, Tasmania, Denny McKenzie & 11 Hughes AD, (1952), Improvements in the Field the plant’s natural profile.12 Associates, P 81, 1990. . Distillation of Peppermint Oil, Corvallis, It is reported that hydro-diffusion 4 Kristiawan M, Sobolik V, Al-Haddad M, Allaf K. Engineering Experimental Station Bulletin No. 31 distillation gives quicker distillations with Effect of pressure drop on the isolation of 1952; Oregon State College, 1-64. lower steam consumption than cananga oil using controlled pressure drop 12 Legast E, Peyron L. Hydrodiffusion Industrial conventional steam distillation.13 However process, Chemical Engineering and Processing Technology to Produce Essential Oils by this process is governed by the physical 2008; 47 (1): 66-75. Steam, in Proceedings of the 11th International laws that govern any other type of 5 Rezzoug SA, Boutekedjiret C, Allaf K. Congress of Essential Oils, Fragrances distillation and the fact that the steam Optimization of operating conditions of rosemary and Flavors Vol. 2 1989; New Dehli, India, travels downwards while cooling may affect essential oil extraction by a fast controlled 69-73. the transfer of latent heat and thus pressure drop using response surface 13 Hall R, Klemme D, Nienhaus J. The H&R Book: increase, rather than decrease distillation methodology, Journal of Food Engineering 2005; Guide to Fragrance Ingredients Vol. 4 1985 time. The tendency to saturate the plant 71 (1): 9-17. London, Johnson Publications, P 13. 0 . July 2012 P E R S O N A L C A R E 73
