Steps in the manufacturing process
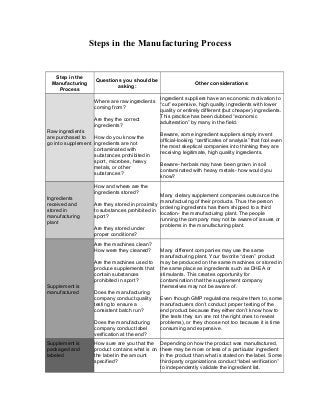
- 1. Steps in the Manufacturing Process Step in the Manufacturing Process Questions you should be asking: Other considerations: Raw ingredients are purchased to go into supplement Where are raw ingredients coming from? Are they the correct ingredients? How do you know the ingredients are not contaminated with substances prohibited in sport, microbes, heavy metals, or other substances? Ingredient suppliers have an economic motivation to “cut” expensive, high quality ingredients with lower quality or entirely different (but cheaper) ingredients. This practice has been dubbed “economic adulteration” by many in the field. Beware, some ingredient suppliers simply invent official-looking “certificates of analysis” that fool even the most skeptical companies into thinking they are receiving legitimate, high quality ingredients. Beware- herbals may have been grown in soil contaminated with heavy metals- how would you know? Ingredients received and stored in manufacturing plant How and where are the ingredients stored? Are they stored in proximity to substances prohibited in sport? Are they stored under proper conditions? Many dietary supplement companies outsource the manufacturing of their products. Thus the person ordering ingredients has them shipped to a third location- the manufacturing plant. The people running the company may not be aware of issues or problems in the manufacturing plant. Supplement is manufactured Are the machines clean? How were they cleaned? Are the machines used to produce supplements that contain substances prohibited in sport? Does the manufacturing company conduct quality testing to ensure a consistent batch run? Does the manufacturing company conduct label verification at the end? Many different companies may use the same manufacturing plant. Your favorite “clean” product may be produced on the same machines or stored in the same place as ingredients such as DHEA or stimulants. This creates opportunity for contamination that the supplement company themselves may not be aware of. Even though GMP regulations require them to, some manufacturers don’t conduct proper testing of the end product because they either don’t know how to (the tests they run are not the right ones to reveal problems), or they choose not too because it is time consuming and expensive. Supplement is packaged and labeled How sure are you that the product contains what is on the label in the amount specified? Depending on how the product was manufactured, there may be more or less of a particular ingredient in the product than what is stated on the label. Some third-party organizations conduct “label verification” to independently validate the ingredient list.
- 2. How sure are you that there is nothing else in the product that is not on the label? Some companies deliberately leave ingredients off of the supplement label, or use made-up names to hide certain ingredients. The FDA has issued many warnings about deliberately spiked or adulterated products. Supplement is distributed Does the supplement company have a list of the sellers of their product so they can recall it if needed? Has the retail outlet made any effort to ensure this product is a properly manufactured and labeled product? Most retailers of dietary supplements are not aware of the various manufacturing or contamination issues of dietary supplements. Just because a product is on a store shelf doesn’t mean it is safe. There are plenty of products that contain substances prohibited in sport at your local grocery store. Retailers are supposed to remove products from shelves when there is a recall, but not all do, and some are quicker than others. Some retailers may not even hear about a recall. You buy the supplement. Do you really know what is in this product? Will you hear about recalls or other enforcement actions against this company if they occur? Regardless of issues such as contamination or mislabeling, some products list right on the ingredients substances that are otherwise prohibited in sport. It is important to be aware of these ingredients just as if they were pharmaceutical drugs. Manufacturers are supposed to report "adverse events" to FDA and these reports are good to review along with public recalls and warnings that they make, but it is wise to remember not all adverse events get reported.
